DNA modification mechanism necessary for individuality of neurons and normal dendrite development
Clustered Protocadherins (cPcdh) are involved in the individuality of neurons.
A group of 13 researchers from the following institutes: the Graduate School of Frontier Biosciences (Osaka University), The Institute of Medical Science (The University of Tokyo), the National Institute for Physiological Sciences (National Institutes of Natural Sciences), and the Center for Developmental Biology (Riken) have discovered that DNA modification during early embryonic stages is related to the individuality of neurons and normal dendrite development.
It was thought for a long time that neurons form elaborate neural circuits through postnatal nerve action. This group discovered that epigenetic regulation during early embryonic stages is important in determining the individuality of neurons and in the forming of nerve circuits and that DNA methyltransferase Dnmt3b was the key regulatory factor. They developed a new method to create chimeric mice by making induced pluripotent stem cell (iPS cells) using DNA modification enzyme-deficient mice.
This group has clarified a molecular mechanism behind the individuality of neurons and formation of nerve circuits. This is expected to contribute to the clarification of causes of certain human genetic and mental diseases.
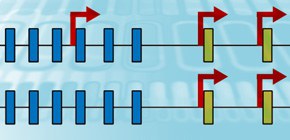
The brain forms complicated nerve circuits and conducts high level information processing. Recently physiologic analysis clarified that individual neural cells have their own circuit characteristics and unique behavior; however, the molecular mechanism for the individuality of neurons and the formation of nerve circuits has been unknown. Clustered Protocadherins (cPcdh) consist of 58 kinds of membrane protein and individual neurons have different cPcdh gene expressions so cPcdh is thought to be involved in the individuality of neurons.
Previous research reported that individual cPcdh genes had their own promoters and gene expression was correlated with DNA methylation states in promoter regions in cultivated cell lines; however, the mechanism in vivo has been unknown. This group analyzed the regulation mechanism for DNA methylation states of cPcdh genes in vivo. As a result, from the 3th to 9th fetal month in which major neurons were produced, transmethylation enzyme Dnmt3b was found to form a different methylation in individual cells.
This group made chimera mice by making iPS cells from Dnmt3b-deficient mice and injecting them into wild-type embryos. These mice were alive after birth and neurons without Dnmt3b were found in the brain. The analysis of the states of cerebellar Purkinje cells revealed that more overlapping dendritic branches in Dnmt3b-deficient cells than in wild-types, which meant Dnmt3b was essential for dendrite formation. They examined the expression of cPcdh genes in individual Purkinje cells by gene expression analysis using fluid integrated circuits. While different cPcdh genes expressed in individual wild-type Purkinje cells, the expression rate of cPcdh genes increased in individual Dnmt3b-deficient Purkinje cells.
CPcdh genes are known to be selected and expressed by a specific enhancer. This group has clarified that Dnmt3b-dependent DNA methylation regulates random expressions of enhancer-dependent cPcdh genes in individual neurons and plays an important role in the individuality of neurons and neural circuit formation.
Research Group Leaders
• Graduate School of Frontier Biosciences (Osaka University): TOYODA Shunsuke , Specially Appointed Researcher, YAGI Takeshi , Professor
• The Institute of Medical Science (The University of Tokyo ): NAKAUCHI Hiromitsu , Professor
• National Institute for Physiological Sciences (National Institutes of Natural Sciences): YOSHIMURA Yumiko , Professor, HIRABAYASHI Masumi , Associate Professor
• Center for Developmental Biology (Riken): OKANO Masaki , Team leader
Abstract
In the brain, enormous numbers of neurons have functional individuality and distinct circuit specificities. Clustered Protocadherins (Pcdhs), diversified cell-surface proteins, are stochastically expressed by alternative promoter choice and affect dendritic arborization in individual neurons. Here we found that the Pcdh promoters are differentially methylated by the de novo DNA methyltransferase Dnmt3b during early embryogenesis. To determine this methylation’s role in neurons, we produced chimeric mice from Dnmt3b-deficient induced pluripotent stem cells (iPSCs). Single-cell expression analysis revealed that individual Dnmt3b-deficient Purkinje cells expressed increased numbers of Pcdh isoforms; in vivo, they exhibited abnormal dendritic arborization. These results indicate that DNA methylation by Dnmt3b at early embryonic stages regulates the probability of expression for the stochastically expressed Pcdh isoforms. They also suggest a mechanism for a rare human recessive disease, the ICF (Immunodeficiency, Centromere instability, and Facial anomalies) syndrome, which is caused by Dnmt3b mutations.

Figure 1
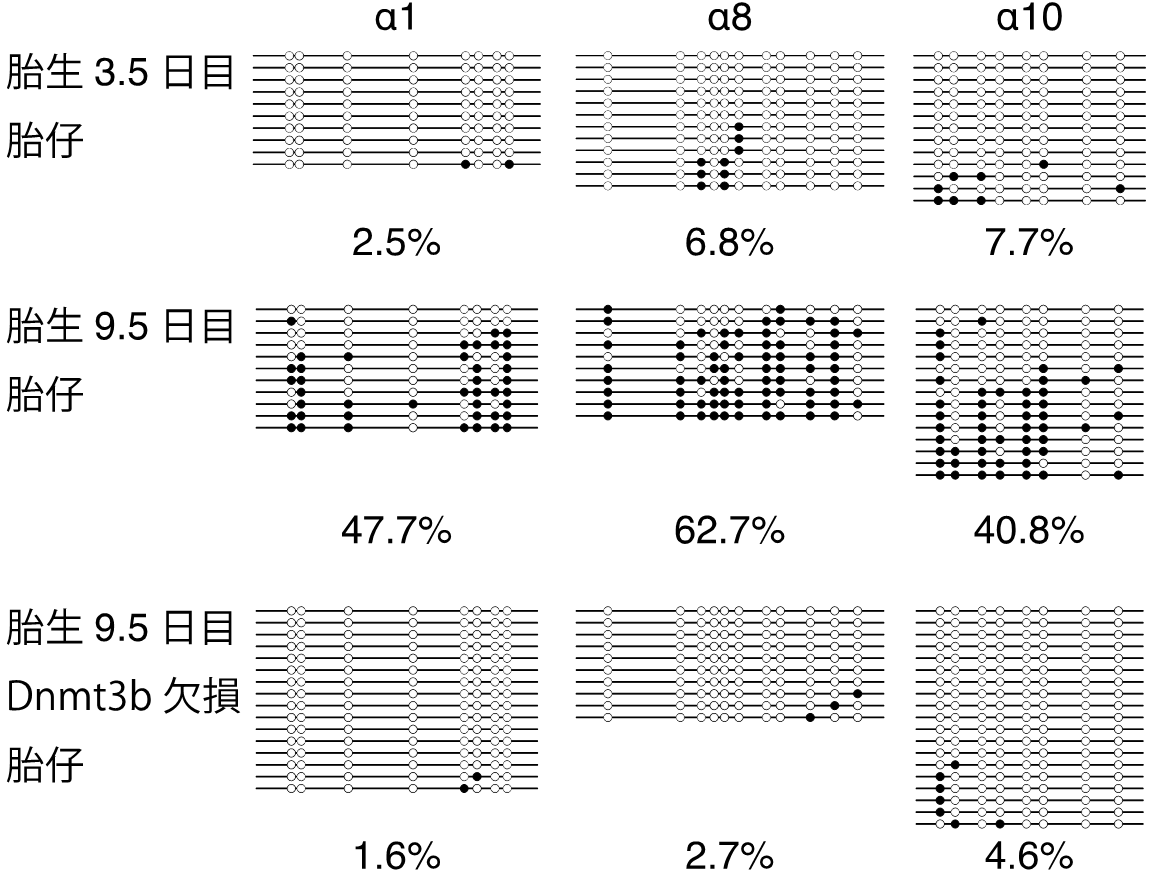
Figure 2
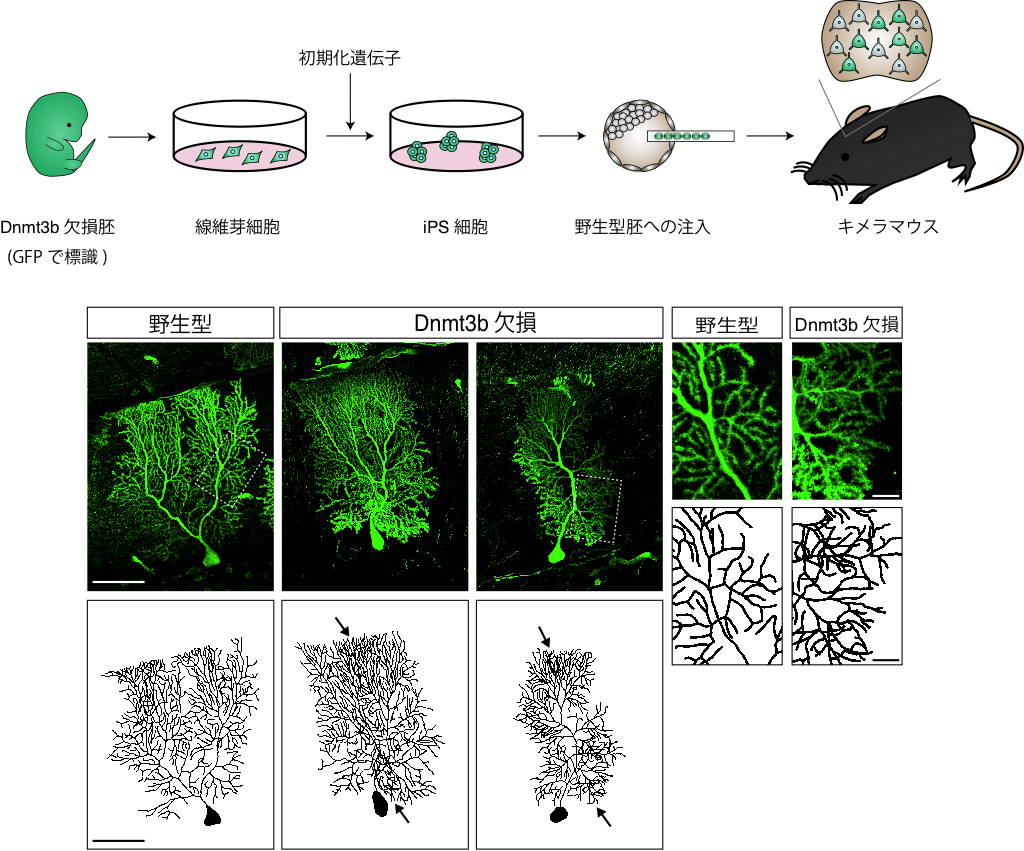
Figure 3

Figure 4

Figure 5
To learn more about this research, please read the full research report entitled " Developmental epigenetic modification regulates stochastic expression of clustered Protocadherin genes, generating single neuron diversity " at this page of the Neuron website.
Related link :
