Discovery of partial action mechanism of drugs for treating treatment-resistant schizophrenia
Will lead to the development of biomarker for predicting response to therapy
Clozapine (CLZ), a gold standard drug for managing treatment-resistant schizophrenia (TRS), is effective in about 60 percent of patients with the disorder; however, potentially life-threatening clozapine-induced agranulocytosis is observed in 0.5 ~ 1 percent of patients in clozapine-treatment. The mechanism of clozapine’s efficacy was still unknown. In order to clarify this mechanism, it was important to conduct research using nerve cells of TRS patients, but research didn’t advance due to the absence of established animal models of TRS.
A group of researchers led by HASHIMOTO Ryota, United Graduate School of Child Development, Osaka University; Associate Professor NAKAZAWA Takanobu, Graduate School of Dentistry, Osaka University; and Professor OKANO Hideyuki, Graduate School of Medicine, Keio University, performed research in order to clarify the molecular mechanism behind differences in response to clozapine therapy in monozygotic twins.
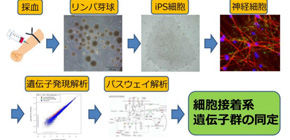
This group produced iPS cell-derived differentiated neurons using immortalized lymphocytes from TRS monozygotic twins with different responses to clozapine and differentiated them into neurons. Extracting all RNA from the obtained iPS cell-derived differentiated neurons, this group conducted gene expression analysis. It was observed that the expression levels of a total of 167 genes were higher in the CLZ-responder compared with those in the CLZ-non-responder. Similarly, the expression levels of a total of 95 genes were lower in the CLZ-responder compared with those in the CLZ-non-responder. An analysis of the 262 genes revealed an enriched functional group in cell adhesion. There were no similar differences in expression levels of genes in immortalized lymphoblasts from patients that were used for producing iPS cells and cell-derived differentiated neurons. Differential gene expression in patients was observed in iPS cell-derived differentiated neurons alone.
In order to clarify molecular mechanism related to response to clozapine, it’s important to confirm reproducibility by using samples from other patients; however, this study suggests that differences in adhesion-related gene expression may be a molecular mechanism behind response to clozapine therapy in patients, and that will become a marker for predicting response to clozapine. Furthermore, this will lead to the development of new effective drugs for treating TRS with less adverse effects. As it is possible to predict response to therapy by using patient-derived neurons, this will also be helpful in making an objective diagnosis.
Abstract
Schizophrenia is a chronic psychiatric disorder with complex genetic and environmental origins. While many antipsychotics have been demonstrated as effective in the treatment of schizophrenia, a substantial number of schizophrenia patients are partially or fully unresponsive to the treatment. Clozapine is the most effective antipsychotic drug for treatment-resistant schizophrenia; however, clozapine has rare but serious side-effects. Furthermore, there is inter-individual variability in the drug response to clozapine treatment. Therefore, the identification of the molecular mechanisms underlying the action of clozapine and drug response predictors is imperative. In the present study, we focused on a pair of monozygotic twin cases with treatment-resistant schizophrenia, in which one twin responded well to clozapine treatment and the other twin did not. Using induced pluripotent stem (iPS) cell-based technology, we generated neurons from iPS cells derived from these patients and subsequently performed RNA-sequencing to compare the transcriptome profiles of the mock or clozapine-treated neurons. Although, these iPS cells similarly differentiated into neurons, several genes encoding homophilic cell adhesion molecules, such as protocadherin genes, showed differential expression patterns between these two patients. These results, which contribute to the current understanding of the molecular mechanisms of clozapine action, establish a new strategy for the use of monozygotic twin studies in schizophrenia research.
To learn more about this research, please view the full research report entitled “ Differential gene expression profiles in neurons generated from lymphoblastoid B-cell line-derived iPS cells from monozygotic twin cases with treatment-resistant schizophrenia and discordant responses to clozapine ” at this page of the Schizophrenia Research website.
Related links
