
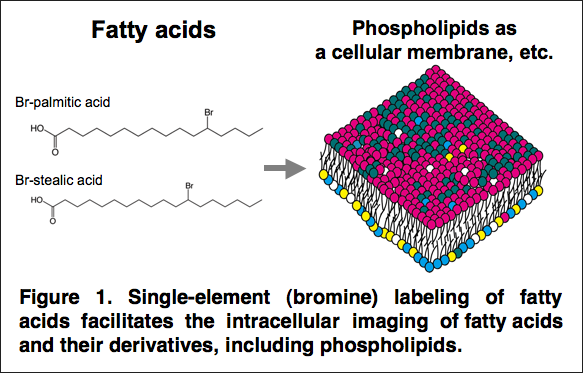
Intracellular imaging of fatty acids by scanning X-ray fluorescence microscopy
Visualization of intracellular localization of fatty acids clarifies changes in lipid metabolism
Fatty acid metabolism abnormality is closely involved with many diseases, so many biochemical research results have been reported. In such a setting, intracellular imaging of fatty acids was thought difficult to achieve because of the difficulty in labeling small molecules of fatty acid. In actuality, labeling big fluorescence molecules greatly inhibits fatty acid metabolism. Observation of unlabeled cells with mass spectrometry or Raman microscope was beneficial, but it was difficult to observe them at the cellular or organelle level.
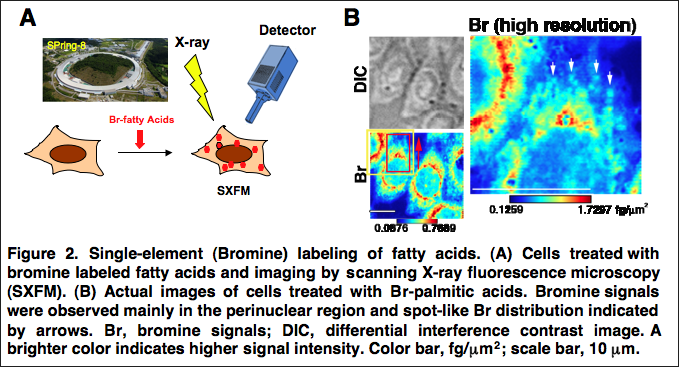
A joint research group of researchers at The University of Tokyo Graduate School of Medicine, Centre national de la recherche scientifique (CNRS), Osaka University Graduate School of Engineering, RIKEN Spring-8 Center, Waseda University Department of Applied Chemistry, and the Research Institute National Center for Global Health and Medicine developed a method for labeling fatty acids with bromine (Br) and applied scanning X-ray fluorescence microscopy (SXFM) at the large synchrotron radiation facility Spring-8. This is the first method for achieving high-resolution imaging of intracellular fatty acids.
In the mice-derived CHO-K1 cells used in this study, labeled element signals from cells were observed around a cell nucleus, exhibiting a distribution similar to that of endoplasmic reticulum and Golgi body. Many enzymes to synthesize intercellular fatty acids were common in that area, so it was expected that many fatty acids would be observed.
Major cell toxicity was not observed in the course of the experiment. In the biochemical study, it was clarified that most element-labeled fatty acids taken up by cells were metabolized into phosphatide. From this, it is thought that the obtained element distribution was that of intracellular-labeled phosphatide. This group also succeeded in visualizing the spot-like distribution of Br of about 1μ in diameter in the cytoplasm with a resolution of some 250 nm, a world first. SXFM can visualize intracellular labeled fatty acid structures sized 1μ or less (the same size as a mitochondrion), providing the organelle-level resolution.
It will become possible to clarify changes in lipid metabolism through visualization of intracellular localization at an organelle level by performing single-element labeling on various fatty acids. It is expected that the use of SXFM will develop research on various life phenomena and various disorders, including inflammation, vascular impairment, and neurological disorders.
Abstract
Fatty acids are taken up by cells and incorporated into complex lipids such as neutral lipids and glycerophospholipids. Glycerophospholipids are major constituents of cellular membranes. More than 1000 molecular species of glycerophospholipids differ in their polar head groups and fatty acid compositions. They are related to cellular functions and diseases and have been well analyzed by mass spectrometry. However, intracellular imaging of fatty acids and glycerophospholipids has not been successful due to insufficient resolution using conventional methods. Here, we developed a method for labeling fatty acids with bromine (Br) and applied scanning X-ray fluorescence microscopy (SXFM) to obtain intracellular Br mapping data with submicrometer resolution. Mass spectrometry showed that cells took up Br-labeled fatty acids and metabolized them mainly into glycerophospholipids in CHO cells. Most Br signals observed by SXFM were in the perinuclear region. Higher resolution revealed a spot-like distribution of Br in the cytoplasm. The current method enabled successful visualization of intracellular Br-labeled fatty acids. Single-element labeling combined with SXFM technology facilitates the intracellular imaging of fatty acids, which provides a new tool to determine dynamic changes in fatty acids and their derivatives at the single-cell level.
To learn more about this research, please view the full research report entitled “ Imaging of intercellular fatty acids by scanning X-ray fluorescence microscopy ” at this page of the FASEB Journal website.
Related links
- X-ray Microscope Team, Graduate School of Engineering, Osaka University (link in Japanese)
- National Center for Global Health and Medicine (link in Japanese)
