Speeding up the journey towards clean energy through photocatalyst optimization
Researchers from Osaka University link the activity of water splitting catalysts to easy-to-measure parameters to enable rapid screening of clean energy generating materials
Photocatalysts that harness light energy and use it to split water into hydrogen and oxygen attract significant scholarly attention, owing to the appeal of hydrogen as a potential clean energy source. However, the optimization of photocatalyst candidate materials usually requires a considerable time investment. Now, researchers at Osaka University have demonstrated a link between easy-to-measure quantities and catalyst performance that could provide a rapid evaluation method.
The conversion of light energy to chemical energy using photocatalysts has been widely reported; however, the continual optimization of photocatalytic materials is critical for their successful application. The properties of photocatalysts, including their surface area, crystallinity, and various electronic features, affect their activity. These properties can be influenced by the techniques and specific conditions used to prepare them, thus leading to a broad range of materials that could be evaluated.
Experiment setup and test of every generated material is a time-consuming step in the development process that has yet to be accelerated—until now. In a report published in ACS Energy Letters , Osaka researchers have shown the relationship between time-resolved microwave conductivity (TRMC) measurements and the photocatalytic performance of semiconductor materials. TRMC is a facile process that allows photocatalysts to be evaluated in powder form, which leads to significantly higher throughput.
“We have been able to show that the oxygen evolution rate of a photocatalyst—which is a measure of activity—can be determined from the photoconductivity and the half-lifetime determined by TRMC,” explains study lead author Hajime Suzuki. “Applying this relationship to materials makes evaluating their potential much more efficient.”
The researchers used their findings to determine the optimum processing temperature for a material that had not been extensively studied, PbBiO 2 Cl, and were able to produce an analogue that had an apparent quantum efficiency of 3%—3 times higher than had been achieved in previous studies using higher processing temperatures.
“We hope that the principles of our findings can be widely applied to improve the efficiency and ease of screening materials, finding candidates, and choosing synthesis conditions,” study corresponding author Akinori Saeki explains. “In terms of the broader picture, high throughput processes could accelerate the development of cleaner energy solutions.
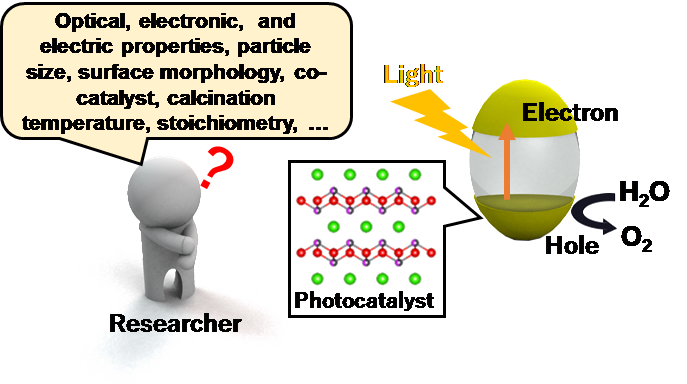
Fig. 1 (Right) Schematic of photocatalytic reaction. (Left) Many factors that affect the photocatalytic performance.
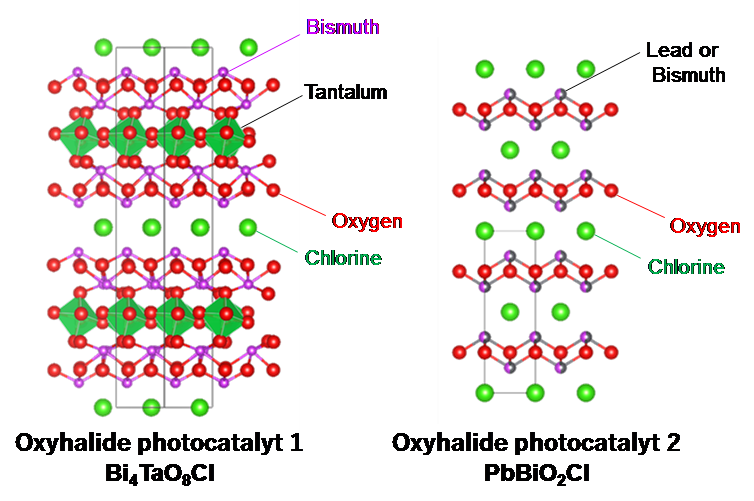
Fig. 2 Crystal structure of oxyhalide photocatalyts 1 and 2.

Fig. 3 (a) Product of microwave photoconductivity intensity and its lifetime of oxyhalide photocatalyt 2 vs calcination temperature. The microwave measurements suggest the optimal temperature of 600 ℃ that is 100 ℃ lower than the reported one. (b) O 2 -evolution rate of oxyhalide photocatalyt 2 prepared by different calcination temperature.
The article, “Photoconductivity−Lifetime Product Correlates Well with the Photocatalytic Activity of Oxyhalides Bi 4 TaO 8 Cl and PbBiO 2 Cl: An Approach to Boost Their O 2 Evolution Rates” was published in ACS Energy Letters at DOI: https://doi.org/10.1021/acsenergylett.9b00793 .
Related links
