Mechanism of familial Parkinson’s disease clarified in fruit fly model
Japanese research collaboration uses fruit flies to show how genetic mutation linked to familial forms of Parkinson’s leads to key events in the disease
Parkinson’s disease (PD) is the second most common neurodegenerative disorder, with up to 10 million people affected worldwide. PD is characterized by progressive increase in movement disability and impaired balance, which are resulted from the loss of dopaminergic neurons in the substantia nigra of the midbrain. While most PD occurs sporadically, about 15% of cases are hereditary and linked to genetic mutations. Although the motor symptoms partly respond dopamine replenishment therapy, there is still a need to develop disease-modifying therapy able to delay or prevent illness.
In a study published in Human Molecular Genetics , researchers from Osaka University (Osaka, Japan) and Tohoku University (Sendai, Japan) have now clarified how a mutation in the DNAJC13 gene may drive the onset of a hereditary form of PD known as PARK21.
The neuropathological hallmark of PD is the loss of dopaminergic neurons with the appearance of Intraneuronal protein aggregates, called Lewy bodies, in affected neurons. Lewy bodies are mainly composed of α-synuclein (αSYN), and much of the research around PD has focused on how this protein contributes to the disease. Prior studies with autopsied brain had revealed that patients with DNAJC13 mutation showed αSYN-positive Lewy pathology in the brainstem, leading the authors to speculate that the DNAJC13 might be able to drive αSYN accumulation in this particular type of PD.
“Mutations in the DNAJC13 gene are known to cause defects in intracellular transport,” study first author Shun Yoshida explains. “A significant amount of αSYN is transported as cargo within neurons, so we speculated that disrupting DNAJC13 might affect the transport of αSYN within neurons. When we exogenously induced mutant DNAJC13 in neuronal cells, we found that cargo trafficking in the cell becomes highly congested, and αSYN is no longer transported correctly. This congestion led us to believe that αSYN might accumulate in neurons, possibly contributing to the initiation and/or progression of the disease.”
The researchers confirmed this hypothesis in the fruit fly Drosophila melanogaster , where genetic mutants can be easily introduced and their effect on brain function readily measured. In this case, the team introduced a human copy of the mutant DNJAC13 , along with human αSYN gene ( SNCA ), allowing them to test whether the defects in cell transporttranslated into problems with motor function in a living organism.
The team found that expressing mutant DNAJC3 with αSYN in fly brains enhanced the accumulation of insoluble, toxic αSYN species, and resulted in a notable loss of dopaminergic neurons. These defects had a significant impact on the motor performance of flies—those with defective DNAJC13 had much greater difficulty carrying out motor tasks such as climbing.
“Our findings suggest mutant DNAJC13 leads to accumulated αSYN, which may lead to Lewy body formation and subsequent damage to motor neurons and pathologies resembling PD,” co-author Takafumi Hasegawa says. “While this work was conducted in Drosophila , we hope that these insights will move us toward a better understanding of how this mutation can lead to PD and provide clues to identify the molecular targets for potential therapeutic interventions.”

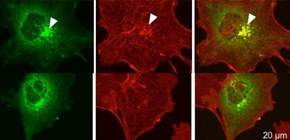
Fig .1 Schematic illustration of the mechanistic link between pathogenic DNAJC13 mutation and αSYN accumulation toward neurodegeneration. The over-expression of PD-linked mutant DNAJC13 resulted in the aberrant accumulation of αSYN in the endosomal compartment, presumably due to defective cargo trafficking from the early endosome to the late and/or recycling endosome, which may provoke cytotoxicity and dopaminergic cell loss that leads to PD. (credit: Tohoku University)
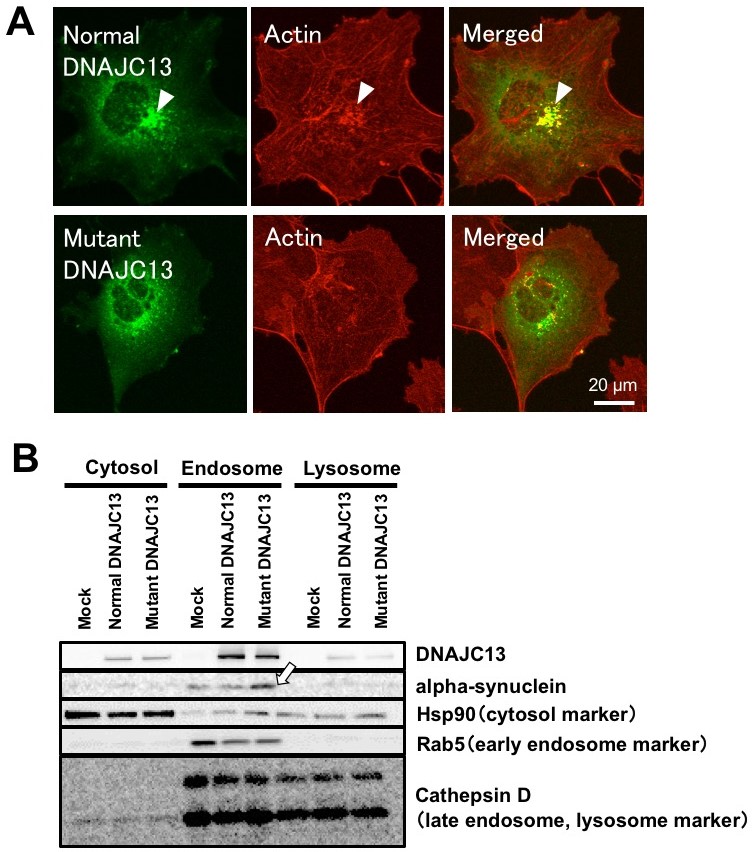
Fig.2 Mutant DNAJC13 impairs cargo transport from early to recycling endosomes. A: Mutant DNAJC13 exhibited less colocalization with F-actin compared with wt DNAJC13, indicating Mutant DNAJC13 disrupts actin cytoskeletal organization on endosomal surface. B: Aberrant retention of αSYN in the endosomal compartment was observed in cells expressing mutant DNAJC13 compared with mock or wt DNAJC13 transformants. (credit: Tohoku University)

Fig.3 Mutant DNAJC13 aggravates αSYN-related phenotype in transgenic flies. In vivo experiments using human αSYN transgenic flies showed that PD-linked mutant DNAJC13 not only increased the amount of insoluble αSYN in fly heads but also induced rough eye phenotype (A) and loss of dopaminergic neurons in fly brains (B). (credit: Tohoku University)
To learn more about this research, please view the full research report entitled "Parkinson’s disease-linked DNAJC13 mutation aggravates alpha-synuclein-induced neurotoxicity through perturbation of endosomal trafficking" at this page of Human Molecular Genetics .
Related link
