Mechanism of cork taint in wine clarified
trichloroanisole (TCA) suppresses the ability to smell
A group of researchers led by Assistant Professor Yuko TAKEUCHI and Professor Takashi KURAHASHI (Graduate School of Frontier Biosciences, Osaka University) and Dr. Hiroyuki KATO (Daiwa Can Company) found that 6- trichroloanisole (TCA), the cause of cork taint [ bouchonné in French] in wine, suppresses olfactory signal transduction.
Even if an ultralow volume of TCA is in the wine, humans sense a deterioration of taste. Wine that has been "corked" is not marketable and is disposed of. The economic loss due to cork taint is estimated to be as much as 1 trillion yen a year; however, the TCA mechanism behind cork taint has been unknown.
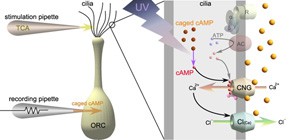
This group administered TCA to olfactory cells and found that the function of cyclic nucleotide-gated (CNG) channels, a major player in transmitting odor information, was suppressed even at pharmacologically ultralow concentrations. Furthermore, the presence of TCA can be detected not only in wine but also in foods and beverages whose quality has deteriorated. Even a small amount of TCA--below measurable limits--can affect the sense of smell in humans. That such a minute quantity of TCA molecules in the air impairs the apparent smell of the food or beverage is surprising. Economic loss for wine alone is 1 trillion yen annually, so the total economic loss due to TCA must be an astronomical figure.
This group has clarified the molecular mechanism of TCA and how it causes the perceived flavor and taste of food to deteriorate. The group's achievement paves the way for molecular drug design for effective sensory depriviation drugs and channel inhibitors.
Abstract
We investigated the sensitivity of single olfactory receptor cells to 2,4,6-trichloroanisole (TCA), a compound known for causing cork taint in wines. Such off-flavors have been thought to originate from unpleasant odor qualities evoked by contaminants. However, we here show that TCA attenuates olfactory transduction by suppressing cyclic nucleotide-gated channels, without evoking odorant responses. Surprisingly, suppression was observed even at extremely low (i.e., attomolar) TCA concentrations. The high sensitivity to TCA was associated with temporal integration of the suppression effect. We confirmed that potent suppression by TCA and similar compounds was correlated with their lipophilicity, as quantified by the partition coefficient at octanol/water boundary (pH 7.4), suggesting that channel suppression is mediated by a partitioning of TCA into the lipid bilayer of plasma membranes. The rank order of suppression matched human recognition of off-flavors: TCA equivalent to 2,4,6-tribromoanisole, which is much greater than 2,4,6-trichlorophenol. Furthermore, TCA was detected in a wide variety of foods and beverages surveyed for odor losses. Our findings demonstrate a potential molecular mechanism for the reduction of flavor.

Figure 1

To learn more about this research, please read the full research report entitled " 2,4,6-Trichloroanisole is a potent suppressor of olfactory signal transduction " at this page of the Proceedings of the National Academy of Sciences of the United states of America website.
Related link :
