Location, location, location: For potassium channels, it depends on functionality
Researchers from The University of Osaka find that potassium KCNQ2/3 channels, which are important for suppressing brain cell excitability, require normal functionality to ensure their proper cellular localization
Potassium KCNQ2/3 channels are crucial for suppressing the excitability of brain cells, or neurons. When these channels don’t work properly, they can cause specific types of epilepsy like benign familial neonatal convulsions and early infantile epileptic encephalopathy.
In a study published recently in PNAS, Japanese researchers have revealed the relationship between KCNQ2/3 channel functionality (i.e., how well they work to control electrical signals in neurons) and localization (i.e., where they are found inside a cell), with important implications for the treatment of these epileptic disorders.
For KCNQ2/3 channels to work properly in the brain, they must have full functionality and be located in the correct cellular region – specifically the axon initial segment (AIS), the site in neurons where electric signals are first triggered, controlling nerve cell activity. This led the research team to wonder: does the functionality of KCNQ2/3 channels affect their cellular localization, or are the two not linked at all?
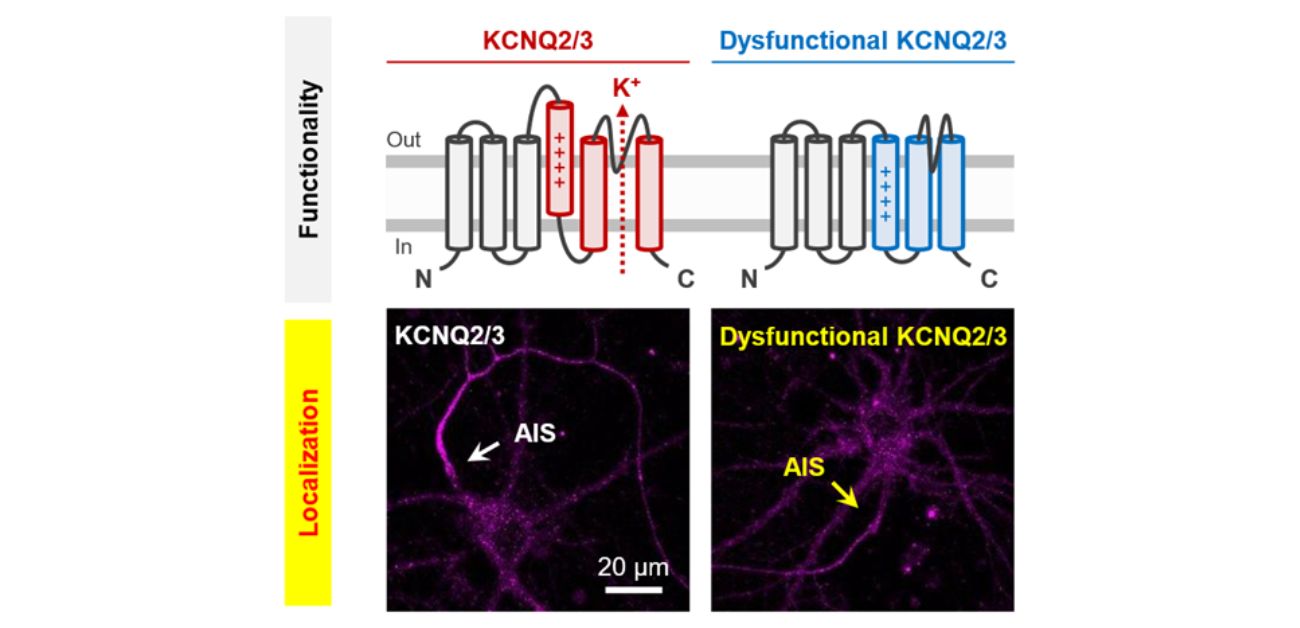
To investigate this potential association, the research team first genetically engineered the functionality of the channels, and then used channel trafficking imaging to visualize whether the channels were taken to their location in the AIS. In this way, they demonstrated that KCNQ2/3 functionality was indeed linked to its trafficking to the correct cellular localization. What’s more, when they used single-molecule imaging, they could see that reduced KCNQ3 functionality actually reduced the AIS localization of KCNQ2/3 by altering the entire trafficking pathway.
“Because we already knew that the localization of KCNQ2/3 to the AIS is regulated by a protein known as ankyrinG, or ankG, we next decided to explore the interactions between full-length KCNQ3 and ankG,” explains lead author of the study Daisuke Yoshioka. “We found that the active conformation of KCNQ3 was essential for its stable binding to ankG, further confirming that functional KCNQ2/3 is needed to ensure its proper accumulation at the AIS.”
Together, these findings highlight the mechanisms underlying the important link between KCNQ2/3 functionality and localization, and provide clues about how their alterations might affect neuronal excitability.
“Now that we know that KCNQ2/3 functionality is closely linked to its localization at the AIS, we have a more concrete target for studying disorders involving altered KCNQ2/3 channels,” says Yasushi Okamura, senior author of the study. “Keeping these potassium channels functioning properly may also be important for ensuring they reach the proper location in brain cells.”
Given that KCNQ2/3‑related neurological disorders, including epilepsy, remain relatively poorly understood and can be difficult to treat, these findings have important implications. They may contribute to the development of new therapeutic strategies that improve the quality of life for young patients and their families.
Fig. 1
Caption: Coupling of KCNQ2/3 functionality to localization. We demonstrated that while normal KCNQ2/3 (left) is strongly enriched at the AIS, dysfunctional KCNQ2/3 (right) shows a significant reduction in AIS localization efficiency. The arrow indicates the position of AIS. Scale bar, 20 µm.
Credit: Daisuke Yoshioka et al., 2026, Coupling of Functionality to Trafficking of KCNQ2/3 Potassium Channels at the Axon Initial Segment, Proceedings of the National Academy of Sciences
Fig. 2
Caption: Binding kinetics of KCNQ2/3 to ankG depend on channel functionality. KCNQ2/3 with normal functionality binds more stably to ankG. This promotes exocytic insertion of KCNQ2/3 at the AIS surface while suppressing its endocytic internalization. In contrast, reduced KCNQ2/3 functionality impairs its binding to ankG, leading to decreased insertion and enhanced internalization at the AIS surface.
Credit: Daisuke Yoshioka et al., 2026, Coupling of Functionality to Trafficking of KCNQ2/3 Potassium Channels at the Axon Initial Segment, Proceedings of the National Academy of Sciences
Fig. 3
Caption: Single-molecule imaging of Halo-TAC-KCNQ3 and ankG-SNAP in the HEK293T cell. Time-lapse TIRFM images depict single molecules of Halo-TAC-KCNQ3 labeled with TMR (green) and ankG-SNAP labeled with 647-SiR (magenta) on the surface of the HEK293T cell. The images are displayed at intervals of 33 ms. Time, s. Scale bar, 5 µm.
Credit: Daisuke Yoshioka et al., 2026, Coupling of Functionality to Trafficking of KCNQ2/3 Potassium Channels at the Axon Initial Segment, Proceedings of the National Academy of Sciences
Notes
The article, “Coupling of Functionality to Trafficking of KCNQ2/3 Potassium Channels at the Axon Initial Segment,” was published in PNAS at DOI: https://doi.org/10.1073/pnas.2527749123
