Seeing the world’s smallest universal joints
Researchers at Osaka University used electron cryomicroscopy to solve the structure of an essential component of the bacterial flagellum with unprecedented resolution
Researchers at Osaka University unveiled the most complete picture of an essential component of the bacterial flagellum to date, the “universal joint” used for transmitting rotary power. This research may help scientists design new antimicrobial agents that target this critical structure, or biomimetic self-propelled nanomachines inspired by the natural system.
Many bacteria have the ability to propel themselves toward food and away from predators using a whip-like “tail” called the flagellum. These flagella are surprisingly complex rotating machines, consisting of several interoperative assemblies and culminating in the long filament propeller outside the cell. The energy for the rotation originates in the basal body inside the cytoplasm, which serves as a motor. However, to consistently transfer torque to the filament with ever-changing orientation, a special mechanism is required, known in the engineering field as a “universal joint.” This is a coupling device that connects rigid rods even when their axes are not always pointing in the same direction. Bacteria have evolved highly efficient part call the hook that acts as a minuscule universal joint much smaller than those we can fabricate.
Of course, scientists have long wondered about the exact structure of flagella, either to copy the design for futuristic robots that can move autonomously around the human body, or else for targeted attacks on bacteria with new antibiotic drugs. While most of the flagellum has been imaged using electron microscopes, it has been very difficult to capture with existing methods the fragile hook section, which is known to be a short, helical assembly that forms a supercoiled tubular structure.
Now, researchers at Osaka University have used improved techniques of electron cryomicroscopy (cryoEM) to precisely image this supercoiled section of the flagellum that has been thought extremely difficult to analyze, to a 3.6 Å resolution that allowed atomic model building. CryoEM, which was the subject of the 2017 Nobel Prize in Chemistry, allows rapidly frozen biological samples to be imaged while they are still in their natural configurations. Even so, the new analysis still required particle-level calculations to reconstruct the most likely structure from the data. “It was thought impossible to look into such a high level of detail for the structure of the hook in its functional state, but we succeeded using cryoEM single-particle analysis,” first author Takayuki Kato says.
This work may have very important implications for fighting infections because the motility behavior of bacteria can greatly influence their chances of becoming virulent. “Bacterial motility is deeply involved in pathogenicity, and the revealed structure can lead directly to the development of new antibiotics,” senior author Keiichi Namba explains.

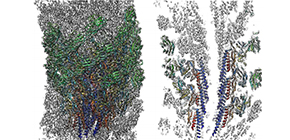
Fig. 1 Three dimensional density map and atomic model of the native supercoiled flagellar hook revealed by cryoEM image analysis. Left: side view; right: a central section.
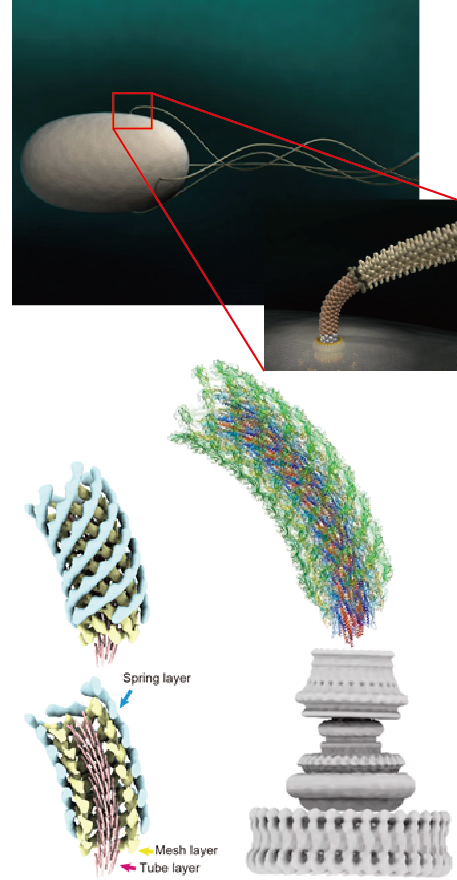
Fig. 2 Computer graphic representation of the bacterial cell and flagella (top left) and its hook connecting the motor and filament (top right). Three-layered tube model of the hook structure (bottom left) and atomic model of the entire hook on top of the flagellar motor (bottom right).
The article, “Structure of the native supercoiled flagellar hook as a universal joint,” was published in Nature Communications at DOI: https://doi.org/10.1038/s41467-019-13252-9 .
Related links
