New Insights into Cause of Phantom Limb Pain May Have Therapeutic Benefits
Link with sensorimotor cortical plasticity revealed
Phantom limb pain is the pain experienced following loss of a limb, either from injury or amputation. This sensation of pain was previously thought to be caused by abnormal plasticity in the sensorimotor cortex of the brain, and rehabilitative therapies have focused on restoring normal motor function to relieve the pain. However, conflicting results left the question of whether and how phantom pain is caused by changes in sensorimotor activity unanswered.
In a new study, researchers based at Osaka University reported on their use of brain–machine interface (BMI) training with a robotic hand on 10 phantom limb patients to investigate the association between changes in symptomatic pain and cortical currents during phantom hand movements. The BMI decodes the cortical signals that instruct the affected hand to move. It then converts this decoded phantom hand movement into movement of the robotic neuroprosthesis. Previous research has shown that BMIs can precisely decode hand movements in real time.
Patients were asked to either open the robotic hand or grasp with it. The cortical currents activated by hand movements were measured using magnetoencephalography (MEG) signals. Results were compared with movement of the intact hand to check that motor information obtained from the sensorimotor cortex was specific. As expected, training with the prosthesis partially restored functioning of the affected hand and increased motor activity in the cortex. However, unexpectedly, participants reported a significant increase in the sensation of pain.
This approach provided a method for inducing localized changes in cortical activity and directly and reversibly studying the relationship with pain.
Indeed, when the patients were asked to move the phantom hand based on MEG signals decoded from movement of the intact hand, cortical sensorimotor activity was disrupted, reducing pain. In contrast with what was previously thought, these findings showed that pain is not reduced by reconstruction of motor function but by changes in cortical plasticity.
Five study participants found that BMI training reduced pain more than previous therapies, suggesting that this is a promising approach for treating phantom limb pain. The training also reduced pain from residual surgery in some patients, therefore may be used to treat other chronic pain conditions.
Abstract
The cause of pain in a phantom limb after partial or complete deafferentation is an important problem. A popular but increasingly controversial theory is that it results from maladaptive reorganization of the sensorimotor cortex, suggesting that experimental induction of further reorganization should affect the pain, especially if it results in functional restoration. Here we use a brain–machine interface (BMI) based on real-time magnetoencephalography signals to reconstruct affected hand movements with a robotic hand. BMI training induces significant plasticity in the sensorimotor cortex, manifested as improved discriminability of movement information and enhanced prosthetic control. Contrary to our expectation that functional restoration would reduce pain, the BMI training with the phantom hand intensifies the pain. In contrast, BMI training designed to dissociate the prosthetic and phantom hands actually reduces pain. These results reveal a functional relevance between sensorimotor cortical plasticity and pain, and may provide a novel treatment with BMI neurofeedback.

Figure 1

Figure 2
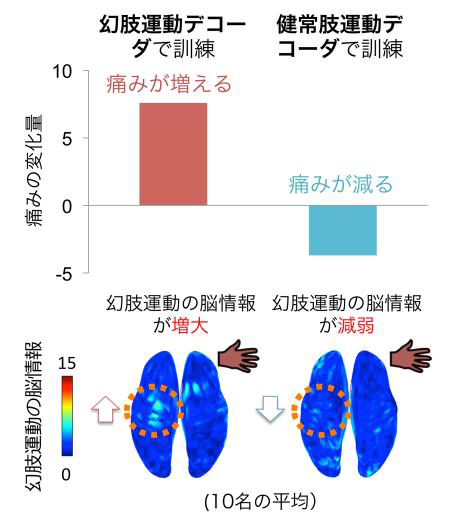
Figure 3
To learn more about this research, please view the full research report entitled “ Induced sensorimotor brain plasticity controls pain in phantom limb patients ” at this page of the Nature Communications website.
Related link
