Important Tricyclic Chemical Compounds with Efficient Chirality Control
Osaka University researchers developed a highly efficient way to produce chiral multi-centered fused tricyclic compounds of which core structure is often found in bioactive compounds including medicines.
Chemists have devoted tremendous efforts to develop a way to build molecules as they want. A single enantiomer (specific special arrangement of atoms) is essential for molecules of specific biomedical properties because of chiral nature of biological receptor sites (usually proteins). A very challenging goal in organic chemistry is asymmetric synthesis, selective preparation of a single enantiomer of a molecule.
Recently, chemists at Osaka University has synthesized highly enantioselective polycyclic chiral compounds with multiple chiral centers in an elegant and selective manner that could open new avenues in research and industry.
In their report in Nature Communications , the Osaka team focused to develop a novel and practical way to synthesize tricyclic molecules, named hydronaphthofurans. These scaffolds are abundant in natural products of important biological activities. The representative members of this family are morphine, azadirachtin, teucvidin and momilacton that are well-known to show a range of biological activities such as analgesic, anti-tumor, insecticides, and allelochemicals (Figure 1). Although some are found in nature, preparing them in the synthetic laboratory with the complete control of selectivity is a major challenge to the synthetic chemists.
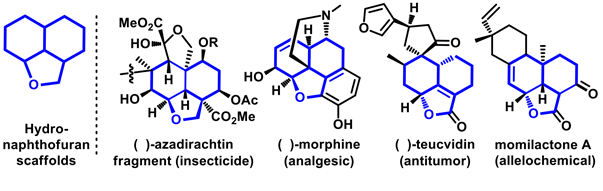
Figure 1. Representative examples of bioactive molecules having hydronaphthofurans scaffolds
A reported synthesis by a Spanish group used an organic molecule based chiral catalyst to produce a tricyclic scaffold with three chiral centers. On the other hand, the Osaka team adopted a conceptually different strategy to build the tricyclic scaffold with five chiral centers (Scheme 1). Starting with a cheap raw material with just one ring (phenols), they used an organometallic nickel-based chiral catalyst to construct further two rings through a reaction termed oxidative cyclization ( 1 to 2 ). In situ displacement of nickel from an intermediate 2 with another reactive molecule of desired substituents afforded a variety of hydronaphthofurans. The final product is formed as single isomer and that contained at least five chiral centers, distributed among all three rings.
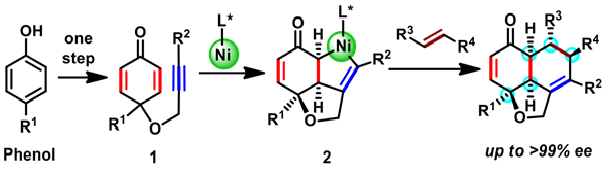
Scheme 1. Two step enantioselective synthesis of hydronaphthofurans scaffolds
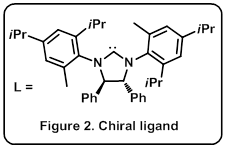
Ravindra Kumar, a one of the team member explains, “The catalyst is key to chiral selectivity, since it is chiral itself.” In fact, although the application of nickel catalysts are widespread in organic synthesis, none had used in this reaction before. “In an organometallic catalyst, it’s crucial to attach a suitable ligand [organic molecule, L* ] to the metal atom to determine the reaction outcome. The ligand is typically a bulky organic molecule with multiple ring systems (Figure 2). In our case, it was a process of informed trial-and-error to find a suitable ligand that gave the desired product as a single compound (out of possible sixteen) both in high yield and with excellent chiral purity.
With the suitable catalyst in hand, the team successfully produced over twenty compounds in good yields and with 94 to 99% enantioselectivity of all five chiral centers – a spectacle achievement! Tolerance of a wide range of substituents (R) (halogens, benzene rings and esters) in the developed method and presence of functionalizable group (carbonyls and olefins) expand the potential synthetic utility of tricyclic products to bioactive molecules after functional group transformation. Apart from offering excellent chiral purity, the reaction is also rapid and economical in terms of starting raw materials, which are crucial requirements for scaling up the research from laboratory to industrial production.
“These tricyclic products have traditionally been made by stepwise ring construction, but that tends to be slow and inefficient,” Sensuke Ogoshi, the team director, says. “Our direct two-step cyclization provides access to a range of very useful scaffolds for natural products. In the long run, this could not only simplify the production of known medicines, but perhaps lead to the discovery of new compounds with medicinal or other biological properties.”
Abstract
Tricyclic furan derivatives with multiple chiral centers are ubiquitous in natural products. Construction of such tricyclic scaffolds in a stereocontrolled, step-economic, and atom-economic manner is a key challenge. Here we show a nickel-catalyzed highly enantioselective synthesis of hydronaphtho[1,8-bc]furans with five contiguous chiral centers via desymmetrization of alkynyl-cyclohexadienone by oxidative cyclization and following formal [4 + 2] cycloaddition processes. Alkynyl-cyclohexadienone was synthesized in one step from easily accessible phenols. This reaction represents excellent chemo-selectivity, regio-selectivity, diastereo-selectivity, and enantio-selectivity (single diastereomer, up to 99% ee). An extraordinary regioselectivity in the formal [4 + 2] cycloaddition step with enones revealed the diverse reactivity of the nickelacycle intermediate. Desymmetrization of alkynyl-cyclohexadienones via oxidative cyclization on nickel was supported by the isolation of a nickelacycle from a stoichiometric reaction. Enantioenriched tricyclic products contain various functional groups such as C=O and C=C. The synthetic utility of these products was demonstrated by derivatization of these functional groups.
To learn more about this research, please view the full research report entitled " Two-step synthesis of chiral fused tricyclic scaffolds from phenols via desymmetrization on nickel " at this page of the Nature Communications website.
Related links
