
Successful fabrication of protein nanowires with a large surface area
Nanowires with aspect ratios of over 1,000
In cooperation with Dr. Atsushi ASANO and Dr. Masaki SUGIMOTO of the Japan Atomic Energy Agency, researchers at Anan National College of Technology and at Tohoku University, a group of researchers at the Graduate School of Engineering, Osaka University* succeeded in fabricating protein nanowires capable of potential use in interactions with living cells as well as in controlling and predicting biological responses.
*Professor Shu SEKI, Professor Takashi HAYASHI, Specially Appointed Researcher Masaaki OMICHI (currently: Anan National College of Technology)
Research on protein nanostructures continues to be conducted because it is thought they can be applied to drug delivery systems, scaffolds for cell adhesion, and diagnosis of diseases. Research on the fabrication of nanostructures by making use of proteins with self-assemble properties has also been conducted. However, such fabrication methods cannot be used for proteins which do not self-assemble; furthermore, it's hard to control the size and homogeneity of nanostructures, an important property in developing drug carriers.
This group succeeded in fabricating extremely thin nanowires of uniform length and thickness by ion beam irradiation of an HSA film and inducing intermolecular cross-linking reactions of the proteins along its trajectory. In conventional processing technologies, the maximum aspect ratio was generally 4 or 5 and it was hard to standardize the length and thickness. However, this group succeeded in fabricating homogeneous protein nanowires of controlled length with a large surface area with aspect ratios of over 1,000.
This group confirmed that enzymes were quantitatively bound to nanowires with large surface areas through interactions between protein molecules or molecules strongly interacting with proteins and that they maintained enzymatic activity. The group also confirmed that it was possible to bond protein nanowires with other organic nanowires fabricated by their method. These findings demonstrate the possibility of selectively fabricating nanowires possessing a variety of chemical compositions and binding them by controlling their length. In fact, it's possible to fully regulate the amount of different enzymes and bind them to the surface of a protein molecule. Nanowires developed with this group's technique promise use in highly sensitive diagnosis of diseases.
Abstract
Protein nanowires exhibiting specific biological activities hold promise for interacting with living cells and controlling and predicting biological responses such as apoptosis, endocytosis and cell adhesion. Here we report the result of the interaction of a single high-energy charged particle with protein molecules, giving size-controlled protein nanowires with an ultra-high aspect ratio of over 1,000. Degradation of the human serum albumin nanowires was examined using trypsin. The biotinylated human serum albumin nanowires bound avidin, demonstrating the high affinity of the nanowires. Human serum albumin–avidin hybrid nanowires were also fabricated from a solid state mixture and exhibited good mechanical strength in phosphate-buffered saline. The biotinylated human serum albumin nanowires can be transformed into nanowires exhibiting a biological function such as avidin–biotinyl interactions and peroxidase activity. The present technique is a versatile platform for functionalizing the surface of any protein molecule with an extremely large surface area.
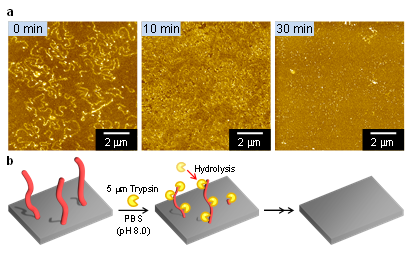
Figure 1

Figure 2
To learn more about this research, please read the full research report entitled" Fabrication of enzyme-degradable and size-controlled protein nanowires using single particle nano-fabrication technique" at this page of Nature Communications website.
Related link:
