A new platform for generating blood cell "seeds" from iPSCs
Production of high-quality hematopoietic progenitor cells (HPCs) using novel extracellular matrix (ECM) proteins
- The research group has optimized a culture method for producing hematopoietic progenitor cells (HPCs), the platform of blood cells, from human induced pluripotent stem cells (iPSCs), and clarified that efficient production of high-quality HPCs can be achieved by adding the growth factor bFGF on them.
- The researchers have developed a new culture substrate P-LM421E8 by fusing laminin-421-E8 fragment with domain 1 of perlecan which has heparan sulfate (HS) chains. They then found that by simply using the P-LM421E8-coated dishes, it is possible to produce high-quality HPCs of the same quality as those obtained with the addition of bFGF without adding bFGF.
- HPCs produced on P-LM421E8 exhibit higher differentiation efficiency and maturity into NK cells than HPCs produced on conventional substrates and are expected to be applied in various areas such as cancer immunotherapy using NK cells.
Outlines
A research team including HEALIOS K.K. and Endowed Chair and Professor Kiyotoshi Sekiguchi of the Institute for Protein Research, the University of Osaka, has developed a new cell culture substrate P-LM421E8, and discovered that it can strongly promote the differentiation of human induced pluripotent stem cells (iPSCs) into hematopoietic progenitor cells (HPCs), which are the "seeds" of blood cells.
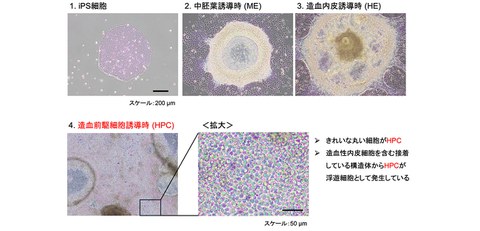
First, the research group conducted a detailed examination of the process of inducing human iPSCs into mesoderm (ME), hematopoietic endothelial (HE), and hematopoietic progenitor cells (HPCs) in a stepwise manner, based on a chemically defined differentiation induction method which had been reported before. (Fig. 1) As a result, it was elucidated that adding basic fibroblast growth factor (bFGF) during both the ME and HE induction phases significantly increased the number of high-quality HPCs that highly expressed CD34 and showed CD117 positive.
Furthermore, when differentiation was induced by coating culture dishes with P-LM421E8 (Fig. 2), which is a fusion of the laminin-421-E8 fragment with domain 1 (D1) of perlecan bearing heparan sulfate (HS) chains, high CD34 expression/CD117-positive HPCs were obtailed at levels comparable to those produced with the addition of bFGF, even without the addition of exogenous bFGF. P-LM421E8 may enhance endogenous FGF signals by retaining FGF in the vicinity of cells via HS chains, and may further enhance the efficiency of intracellular signaling by coordinating signals from integrins, which are P-LM421E8 receptors (Fig. 2).
In addition, when the obtained HPCs were differentiated into natural killer (NK) cells, it was confirmed that the differentiation efficiency into NK cells and the expression of maturation markers were higher than those of HPCs derived from the conventional substrate (LM511E8).
These results will promote the development of regenerative medicine, particularly cancer immunotherapy using iPSCs, and are expected to contribute to future medical treatments as a platform for the stable production of high-quality immune cells.
Fig. 1 Differentiation induction of human iPSCs into hematopoietic progenitor cells (HPCs)
Credit: Kiyotoshi Sekiguchi
Fig. 2 Mechanism of P-LM421E8
Credit: Kiyotoshi Sekiguchi
Research Background
Human iPSCs are pluripotent stem cells (PSCs) that can differentiate into various cells in the body, and are expected to be used in regenerative medicine to replace cells lost due to illness or injury, as well as in the development of new drugs. In particular, the technology to create hematopoietic progenitor cells (HPCs), which are the precursors of blood cells (such as red blood cells and immune cells), from iPSCs is attracting attention as fundamentals for treating various blood diseases and for cancer immunotherapy, which produces large quantities of immune cells that attack cancer. On the other hand, it is not easy to reproducibly and stably produce HPCs that are high-quality and quantitative enough for clinical application without containing animal-derived components.
In the body, early cells equivalent to iPSCs differentiate into ME, which then pass through the HE that produces blood cells, leads to creation of HPCs. This process is known to be coordinated and controlled by multiple signals, such as FGF, Wnt, BMP, and VEGF. Furthermore, in recent years, it has become clear that ECM proteins, which act as scaffolds for cells to adhere, proliferate, and differentiate, are also important factors that greatly affect the maintenance and differentiation efficiency of iPSCs. At the University of Osaka, laminin-421-E8 fragment, the smallest adhesive fragment of laminin is used to enable highly efficient expansion and culture of human ES/iPS cells and the induction of differentiation into a variety of cells, including vascular, neural, and muscle cells. In addition, the research group recently developed a new concept substrate P-LM421E8 in which perlecan D1 is bound to laminin E8 and growth factors such as FGF are held and presented on the scaffold via heparan sulfate (HS) chains. They also have reported that this increases the efficiency of muscle differentiation via paraxial mesoderm.
However, it was unclear how FGF and P-LM421E8 act in the system for inducing HPCs from human iPSCs, and to what extent they affect the quality of HPCs.
Research Contents
● bFGF enhances the quality of HPC, not the quantity
Based on a chemically defined differentiation induction method from previous research, the research group differentiated human iPSCs in the following three stages (Fig. 1).
1. Mesoderm (ME) induction: Add BMP4, Wnt signaling activator, and VEGF
2. Hemogenic endothelial (HE) induction: Add TGF-β signaling inhibitor, SCF, and VEGF
3. Hematopoietic progenitor cell (HPC) induction: Add SCF and FLT3L
During this process, the research group compared the conditions in which bFGF was added to "only the ME induction phase," "only the HE induction phase," and "both," then analyzed the surface markers of floating cells collected on days 14 and 20. As a result, it was found that the percentage of high-quality HPCs that highly expressed CD34 and indicated CD117 positive significantly increased at both timings. They also demonstrated that the highest percentage of HPCs was produced especially when bFGF was added during both periods. On the other hand, the percentage of HE cells (CD34⁺/KDR⁺) assessed on day 4 did not change significantly with or without bFGF, and the total number of floating cells did not increase dramatically with the addition of bFGF. According to these results, it was indicated that bFGF acts not to increase HEs, but rather to enhance the quality and stability of HPCs produced from it.
● The novel substrate P-LM421E8 realizes high-quality HPCs even without bFGF
Next, the research group developed a new culture substrate P-LM421E8, by fusing the laminin-421-E8 fragment with the perlecan D1, and compared it with the conventional substrate, LM511E8. When human iPSCs were induced to differentiate on these substrates under the same conditions, it was found that the percentage of CD34-high expression/CD117-positive HPCs on P-LM421E8 was approximately three times higher than on LM511E8. On the other hand, there was no significant difference in the total number of floating cells collected under both conditions, suggesting that, same as the condition of additing bFGF, P-LM421E8 primarily enhances the quality of HPCs.
Furthermore, to separate the contribution of perlecan D1 from differences in laminin α chains, the researchers prepared LM421E8 without D1, and compared it with P-LM421E8 and LM511E8.
As a result, only P-LM421E8 indicated a significant HPC enhancement effect. This means that the HPC-enhancing effect is not due to differences in laminin itself, but is largely due to perlecan D1 (HS chains).
● The actions of P-LM421E8 and bFGF overlap, and no synergistic effect is seen when used in combination
To examine the relationship between P-LM421E8 and bFGF, the research group compared three conditions: LM511E8 only, LM511E8 + bFGF, and P-LM421E8 + bFGF. As a result, the quality of HPCs produced by P-LM421E8 + bFGF was almost the same level as that produced with LM511E8 + bFGF, and no clear additional effect was observed from the combined use of P-LM421E8 and bFGF. This indicates that P-LM421E8 may be able to capture endogenous FGF secreted by the cells themselves with HS chains and present it in the vicinity of the FGF receptor to induce intracellular signaling activation similar to that induced by the addition of exogenous bFGF (Fig. 2).
● P-LM421E8-derived HPCs have high differentiation potential and maturity into natural killer (NK) cells
To assess the functional quality of HPCs, the produced HPCs were differentiated into natural killer (NK) cells. After 42 days cultivation in a medium containing IL-15, IL-7, SCF, and FLT3L, CD56-positive NK cells accounted for 70-90% or more of the HPC-derived cells produced on P-LM421E8. In contrast, the HPC-derived cells obtained on LM511E8 showed only 6-35% cell viability. Furthermore, analysis of markers related to the maturation and function of NK cells (NKG2A, NKp30, NKp44, and NKp46) showed that P-LM421E8-derived NK cells had high expression of NKp30/NKp44/NKp46, while NKG2A expression tended to be relatively low. This means that P-LM421E8 is effective in producing high-quality HPCs, which also contributes to the maturity and functionality of HPCs when differentiated into NK cells.
Social Impact of Research
This research may have a major impact on future medicine and industry as follows:
● Platform for stable supply of highly functional NK cells
In NK cell therapy, a type of cancer immunotherapy, the development of standardized "off-the-shelf" cell products (which can be used by anyone at any time) is required. As shown in this study, the ability to obtain highly efficient and mature NK cells from P-LM421E8-derived HPCs is important as a fundamental technology for an NK cell creation platform using iPSCs. This is expected to develop more stable and practical NK cell products.
● Design guidelines for next-generation culture substrates based on ECM engineering and signal control
The combination of laminin-421-E8 fragment and perlecan D1 indicated a novel concept of scaffolding growth factors using HS chains. This technology can be applied to promote differentiation and improve maturation of various cell types, not just HPCs and muscle cells. This could contribute to the development of new cell culture materials and regenerative medicine products as a guideline for designing highly functional substrates that combine ECM and signaling factors.
● Construction of chemically defined, safe, and highly reproducible differentiation systems
The differentiation induction system used in this study is completely under chemically defined conditions and does not contain animal-derived components or use serum or feeder cells. Such configuration is suitable for pharmaceutical-level quality control and is characterized by very high stability and reproducibility. As protocols are standardized and scaled up in the future, application to the production of clinical-grade hematopoietic and immune cells will become more realistic. Establishing this safe and highly reproducible differentiation system will be an important key to promoting the widespread use of regenerative medicine and cell therapy.
Notes
The article, “P-LM421E8, the heparan sulfate chain-conjugated laminin-421-E8 fragment, drives differentiation of human induced pluripotent stem cells into hematopoietic progenitor cells comparable to basic fibroblast growth factor in a chemically defined system,” was published in Matrix Biology Plus at DOI: 10.1016/j.mbplus.2025.100188
