\Entering an era where cells are administered through vaccination to fight cancer/CAR-T therapy simply by administration
Developed a new immunotherapy for solid cancers using mRNA vaccine technology
- Developed a new method using mRNA-DDS to create CAR-T cells in the body and discovered that it can completely eliminate solid cancers and induce long-term immune memory.
- Conventional CAR-T therapy had limited efficacy due to challenges such as the instability of target antigens, the difficulty in maintaining CAR activity in an immunosuppressive environment, and the need for complex procedures.
- In this study, researchers focused on FAP, which is highly expressed in CAFs, and enabled the temporary expression of FAP-specific CARs in immune cells in the body using mRNA-LNPs, thereby modifying TMEs and inducing potent antitumor immunity.
- In addition to its potential application as a new CAR immunotherapy for solid cancers which are prone to recurrence, optimization of mRNA modification (m⁶A), MIF-CD74 blockade, and HOX gene regulation will lead to the development of a safe, low-cost next-generation cancer treatment in which CAR-T cells work in the body simply by being administered.
Outlines
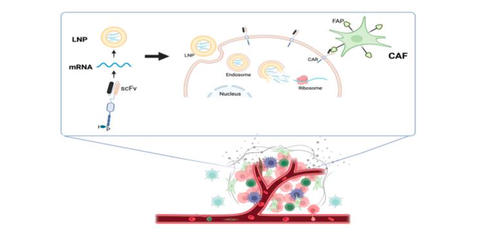
A research group including Specially Appointed Assistant Professor Sikun Meng, Specially Appointed Assistant Professor Tomoaki Hara (full-time), and Specially Appointed Professor Hideshi Ishii (full-time) (Medical Data Science) of the Graduate School of Medicine, the University of Osaka, in collaboration with Saitama Medical University, Tokai University, Science Tokyo, and the University of Copenhagen in Denmark, has revealed that by administering mRNA via lipid nanoparticles (LNPs) to temporarily express FAP-targeted CARs in immune cells in the body, it is possible to completely eliminate solid cancers and induce longer-term antigen-specific immune memory.
In this study, the researchers demonstrated that expressing FAP-targeting CAR, which is specific to cancer-associated fibroblasts (CAFs), a major component of tumor stroma, via mRNA-LNP reconstructs the immunosuppressive tumor microenvironment that conventional CAR-Ts struggle with, achieving complete tumor elimination in multiple solid cancer models, and further demonstrating long-term recurrence prevention (immunological memory) after treatment.
Until now, CAR-T therapy for solid cancers has been thought to have limited efficacy due to the instability of target antigens and the strong immunosuppressive environment, and methods for safely inducing CAR activity in the body have not been elucidated.
In this study, the research group elucidated the mechanism by which FAP-targeting CAR-T are directly induced into immune cells in the body using mRNA-LNPs, thereby not only suppressing solid cancers but also establishing long-term immune memory. This is expected to lead to applications in safe, low-cost in-vivo CAR-T cancer immunotherapy that does not require complex cell manipulation.
Fig. 1 Schematic representation of the mRNA-LNP-based FAP-targeted CAR strategy
Credit: Hideshi Ishii
Research Background
Until now, CAR-T therapy for solid cancers has been known to be less effective due to the instability of tumor-specific antigens and the presence of a highly immunosuppressive tumor microenvironment (TME).
Furthermore, although therapies targeting FAP, which is specifically expressed in cancer-associated fibroblasts (CAFs) that form tumor stroma, were considered promising, they faced the following challenges:
- Conventional cell-manipulated CAR-T have challenges in terms of safety and control
- Maintaining CAR activity within TMEs in solid cancers is difficult
- The antitumor effect is not sustained easily
In addition, in-vivo CAR expression technology using mRNA has been proposed, but aspects such as optimizing stable expression, effectively disrupting tumor stroma, and inducing long-term immune memory have not been fully elucidated. As a result, there are still many challenges to realize in-vivo CAR-T that is therapeutically effective against solid cancers. In order to overcome these challenges, this study was conducted.
Research Contents
The research group elucidated that transient CAR induction targeting tumor stroma (CAF) is extremely effective in suppressing solid cancers by encapsulating mRNA in lipid nanoparticles (LNPs) and administering them to temporarily express FAP-targeting CAR in immune cells within the body.
This demonstrated that by destroying CAFs, which constitute the tumor microenvironment (TME), rather than the tumor cells themselves, the immunosuppression of the overall tumor is released, inducing a powerful anti-tumor immune response. This presents a novel mechanism of action that could achieve complete elimination or suppression of recurrence of solid cancers, which was difficult to achieve with conventional CAR-T therapy.
Furthermore, by demonstrating that chemically-modified mRNA (m⁶A) significantly improves CAR expression efficiency and therapeutic efficacy, and that the MIF–CD74 pathway and HOX transcription factors are novel therapeutic targets involved in enhancing the effects of mRNA-LNP CARs, an optimization strategy is presented for realizing more efficient and sustainable in-vivo CAR-T therapy.
Social Impact of Research
This research suggests the possibility of realizing a new solid cancer immunotherapy that can induce CAR activity in the body simply by administering it, replacing conventional CAR-T therapies that involve complex cell manipulation.
In addition, the technology to control tumor stroma targeting FAP achieved high therapeutic efficacy even against solid cancers that were previously resistant to treatment, It opened the way for sustained cancer management through recurrence suppression and the acquisition of long-term immunological memory.
Furthermore, by utilizing novel targets such as m⁶A-modified mRNA and cytokine regulatory pathways like the MIF-CD74 pathway and HOX transcription factors, the optimization of safer and more powerful in-vivo CAR-T technology will significantly improve its applicability in clinical settings, including expanding the range of treatable cancer types, reducing treatment costs, and enabling treatment initiation in a shorter period of time.
This technology also has higher compatibility with mRNA vaccine technology and is expected to become widespread as a social infrastructure, thus potentially leading to a transformation in standard approaches to cancer treatment.
Notes
The article, “Targeting fibroblast activation protein in solid tumors via LNP-mediated CAR-mRNA delivery promotes durable tumor eradication in murine models,” was published in American journal of Scientific Reports (online) at DOI: https://doi.org/10.1080/23294515.2025.2474928.

