Identified how inflammation worsens fatty liver spreads
Inflammation spreads from macrophages via vascular endothelial cells
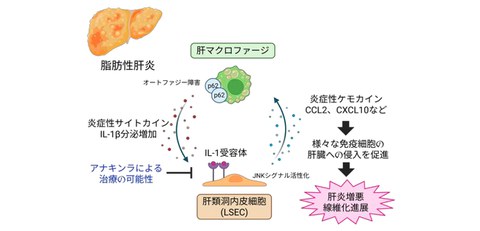
- Autophagy of immune cells (macrophages) in the liver is suppressed in fatty liver, and the inflammatory substance IL-1β is excessively secreted.
- Excessive IL-1β released by macrophages in the liver spreads inflammation via liver sinusoidal endothelial cells (LSECs), which make up the blood vessels of the liver.
- IL-1 receptor antagonist, which suppress IL-1β signaling, may potentially improve hepatitis and liver fibrosis.
Outlines
A research group including Kenji Fukumoto, a graduate student in the Department of Gastroenterology and Hepatology at the Graduate School of Medicine, the University of Osaka (at the time of the research: doctoral student, currently: Attending Staff (Physician) at the University of Osaka Hospital), and Lecturer Hayato Hikita of the same Department, has revealed that the transmission of inflammatory signals between cells around the periportal (near the large blood vessels that enter the liver) plays an important role as one of the causes of worsening fatty liver.
In recent years, due to the Westernization of dietary habits, it is said that one in four people in Japan suffers from fatty liver, making it the leading cause of liver disease. When fatty liver disease worsens and inflammation intensifies, it can develop into Metabolic dysfunction-associated steatohepatitis (MASH), increasing the risk of progression to cirrhosis or liver cancer. However, how this inflammation begins and spreads has not been fully understood until now.
In this study, the research group used a cutting-edge technique called single-cell analysis to examine the livers of mice with fatty liver disease in detail. They discovered the inflammatory substance IL-1β, released by immune cells (macrophages) present in the liver, acts on LSECs, causing them to produce large amounts of substances (chemokines) that intensify inflammation (Fig. 1).
Furthermore, by performing single-cell analysis (spatial transcriptome analysis) that preserves the positional information of cells in the livers of patients with fatty liver disease, it was shown that a similar phenomenon may be occurring in humans, and that inflammation is transmitted from macrophages to LSECs, leading to the worsening of fatty liver disease.
Fig. 1 Mechanism of aggravation of fatty liver disease by intercellular interactions
Credit: Hayato Hikita
Research Background
Fatty liver is a very common disease in Japan, affecting an estimated one in four people. Early-stage fatty liver can be improved with exercise and dietary changes, but once inflammation and fibrosis progress, it becomes difficult to reverse and can lead to liver cancer in the future. However, how fatty liver progresses to MASH has not been fully understood until now. Understanding this mechanism could lead to new treatments to prevent cirrhosis and liver cancer.
Research Contents
The research group created a mouse model that induces fatty liver disease and performed single-cell analysis on the liver. This led to a discovery of the fact that in fatty liver disease, IL-1β is secreted mainly by macrophages, and its receptor, IL-1R1, is highly expressed in LSECs (Fig. 2). Furthermore, autophagy dysfunction was suggested in these macrophages within the liver.
Fig. 2 Single-cell RNA analysis of liver tissue using mice with fatty liver disease
Credit: Hayato Hikita
Cell experiments revealed that fatty acids increase IL-1β secretion by disrupting macrophage autophagy, and that this IL-1β enhances the expression of inflammatory chemokines such as CCL2 and CXCL10 in LSECs (Fig. 3).
Fig. 3 Macrophage-derived IL-1β enhances chemokine expression in LSECs
Credit: Hayato Hikita
Furthermore, administering endothelial cell IL-1R1 knockout (a technique that intentionally disrupts or eliminates the function of a specific gene) or an IIL-1 receptor antagonist (anakinra) to a mouse model of fatty liver disease improved inflammation and fibrosis in liver tissue (Fig. 4).
Fig. 4 Potential of IL-1 receptor-targeted treatment for fatty liver disease
Credit: Hayato Hikita
Furthermore, spatial transcriptome analysis using the livers of patients with fatty liver disease showed that IL-1R1-mediated interactions between macrophages and LSECs are important in humans as well, and that they are particularly active around the periportal in the liver (Fig. 5).
Fig. 5 Analysis of spatial intercellular interactions using liver tissue from patients with fatty liver disease
Credit: Hayato Hikita
Social Impact of Research
This study suggests that treatment targeting IL-1β or the IL-1 receptor may be effective in preventing the aggravation of fatty liver disease. While early-stage fatty liver disease can be improved with changes of daily habits, advanced fatty liver disease, characterized by inflammation and fibrosis, is difficult to treat and is a major cause of future cirrhosis and liver cancer. Therefore, there is a strong demand for treatment that stops the very first step in worsening the disease. The function of IL-1β clarified in this study is an important mechanism that commonly enhances inflammation in various types of hepatitis, including not only fatty liver disease but also viral hepatitis and alcoholic hepatitis, and its application to a wide range of liver diseases can be expected. Furthermore, it was found that in fatty liver disease, periportal of the liver strongly reacts particularly as the starting point of inflammation. This is an important discovery that will lead to the development of indicators for early diagnosis and the selection of more effective treatment targets.
Notes
The article, “Liver Sinusoidal Endothelial Cells Promote Metabolic Dysfunction-associated Steatohepatitis Progression via Interleukin-1R1-mediated Chemokine Production Induced by Macrophage-derived Interleukin-1β,” was published in American scientific journal of Cellular and Molecular Gastroenterology and Hepatology (online) at DOI: https://doi.org/10.1080/23294515.2025.2474928.

