Discovered the mechanism by which ferroptosis is enhanced in diabetic kidneys
Autophagy impairment and AMPK inactivation enhance ferroptosis
- The mechanism by which ferroptosis is enhanced in diabetic kidneys has been elucidated.
- Renal proximal tubule cells from diabetic patients were found to have autophagy impairment and AMPK inactivation, as well as lipid peroxide accumulation, a characteristic of ferroptosis (cell death). It has been revealed that in diabetic kidneys, autophagy impairment leads to the accumulation of mitochondrial ROS, enhancing ferroptosis. Furthermore AMPK-ACC system inactivation also enhances ferroptosis.
- Now that the mechanism by which diabetic patients become vulnerable to acute kidney injury (AKI) has been revealed, it is expected that new treatments will be developed to reduce the risk of AKI in diabetic kidneys and suppress the progression of renal failure.
Outlines
A research group including Sho Matsui (Nephrology), the Attending Staff (Physician) at the University of Osaka Hospital, Specially Appointed Professor Takeshi Yamamoto (Full Time) and Professor Yoshitaka Isaka (the Department of Nephrology) at the Graduate School of Medicine of the University of Osaka, has clarified the mechanism by which ferroptosis is enhanced in diabetic kidneys.
Diabetic patients are known to be at increased risk for acute kidney injury (AKI), but the mechanisms underlying their vulnerability to AKI remain unknown. Recently, it has become clear that ferroptosis, a cell death process characterized by the lipid peroxide accumulation, plays an important role in AKI of renal tubules. However, the mechanism in which whether ferroptosis is (or was) involved or not in AKI vulnerability in diabetic kidneys has not been fully elucidated.
In this study, the research group found that diabetic patients had a lipid peroxide accumulation, a characteristic of ferroptosis, and impaired autophagy, and inactivated AMPK. They also indicated that autophagy impairment leads to the accumulation of mitochondrial ROS and enhances ferroptosis. Furthermore, it was found that AMPK inactivation suppresses the activity of the downstream ACC, thereby enhancing ferroptosis.
Now that the mechanism by which diabetic patients become vulnerable to AKI has been clarified, it is expected to lead to the development of new treatments that suppress ferroptosis in renal tubules.
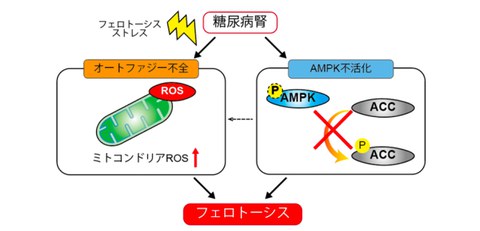
Fig. 1 Research overview
Diabetic kidneys result in autophagy impairment and the AMPK-ACC axis inactivation. Autophagy impairment leads to mitochondrial ROS accumulation and predisposed to ferroptosis. Furthermore, inactivation of the AMPK-ACC axis makes ferroptosis more likely to occur.
Credit: Takeshi Yamamoto
Research Background
Diabetic patients are known to be at increased risk for acute kidney injury (AKI), but the mechanisms underlying their vulnerability to AKI remain unknown. Recently, it has become clear that ferroptosis plays an important role in AKI, however, its mechanism if ferroptosis is or was involved in AKI vulnerability in diabetic kidneys has not been fully elucidated.
The research group therefore hypothesized that ferroptosis is more likely to occur in diabetic kidneys and verified this using human kidney biopsy samples and mouse experiments using an ischemia-reperfusion model.
Research Contents
Using samples from patients who underwent kidney biopsy, it was found that the renal tubular cells of patients with diabetic kidney disease (DKD) exhibited autophagy impairment (accumulation of p62, which is degraded by autophagy), AMPK inactivation, and lipid peroxide accumulation which was assessed with 4HNE (4-hydroxynonenal), a characteristic of ferroptosis (Fig. 2).
Fig. 2 Lipid peroxide, a characteristic of ferroptosis, is accumulated in renal tubular cells of diabetic patients
Renal tubular cells from a control patient (top) and a diabetic patient (bottom)
Credit: Takeshi Yamamoto
Staining of 4HNE (lipid peroxide) is elevated in diabetic patients. The staining level or dot count of p62 (accumulation caused by autophagy impairment) is elevated in diabetic patients. Furthermore, AMPK staining is reduced in diabetic patients.
Next, when ischemia-reperfusion injury was induced in diabetic mouse models (STZ-induced mouse model of type 1 diabetes and the db/db mouse model of type 2 diabetes) using ferrostatin 1, a ferroptosis inhibitor, it was found that ferroptosis was enhanced in both diabetic models.
Also, experiments using autophagy-impairment mice revealed that ferroptosis was enhanced in autophagy-impairment renal tubular cells (Fig. 3). Furthermore, it was found that autophagy-impairment cells had mitochondrial damage and an accumulation of mitochondrial ROS and mitochondrial lipid peroxides. The research group has found that ferroptosis enhancement was suppressed in mitochondria-depleted Rho0 cells, and that ferroptosis was enhanced in autophagy-impairment cells due to mitochondrial ROS accumulation.
Since the research group observed a decrease in both AMPK and ACC in the kidneys of diabetic mice, they investigated whether ferroptosis, which is enhanced in diabetes, could be improved by activating AMPK. In the experiment, AICAR, an AMPK activator was administered to diabetic model mice and evaluated the effect using an ischemia-reperfusion model. Then they found that ferroptosis, which was exacerbated in both type 1 and type 2 diabetic groups, was suppressed by AICAR (Fig. 4).
Fig. 3 Ferroptosis is enhanced in autophagy impairment renal tubules
Credit: Takeshi Yamamoto
Above shows the immunofluorescence staining of wild-type mice 2 days after ischemia-reperfusion injury in the kidneys (left) and proximal tubule-specific Atg5 knockout mice (autophagy-impairment mice) (right). Red: 4HNE, green: p62 (accumulated due to autophagy impairment), purple: megalin. While wild-type mice did not show accumulation of p62 or lipid peroxide (4HNE), autophagy- impairment mice showed accumulation of lipid peroxide in autophagy-impairment tubular cells (tubules with p62 accumulation) and showed enhanced ferroptosis in autophagy-impairment cells.
Fig. 4 AICAR ameliorates diabetic ferroptosis induced by ischemia-reperfusion injury
Credit: Takeshi Yamamoto
Wild-type mice, STZ-induced mouse model of type 1 diabetes, and the db/db mouse model of type 2 diabetes were administered vehicle or AICAR, and the kidneys were subjected to ischemia-reperfusion injury. PAS staining 2 days after administration is shown above. Renal damage worsens in STZ mice and db/db mice, but it is confirmed that AICAR improves renal damage.
Social Impact of Research
The occurrence rate of AKI is high, and it increases the risk of death. It is also known that even if a patient recovers from AKI, they are likely to progress to chronic kidney disease. If AKI is reduced, the risk of death and chronic kidney disease will be reduced, and ultimately leads to the reduction of the need for dialysis.
It is expected that autophagy and AMPK will be activated, and ferroptosis inhibitors will reduce the risk of AKI in diabetic kidneys leading to the development of new treatments that suppresses the progression of renal failure.
Notes
The article, “Defective Autophagy and AMPK Inactivation Drive Ferroptosis in Diabetic Kidney Disease,” was published in Diabetologia at DOI: https://doi.org/10.1080/23294515.2025.2474928.
