Making safe choices: It’s in our DNA
Researchers from Osaka University demonstrate how DNA replication machinery plays an important role in the choice of recombination pathway at the centromeres of chromosomes to limit harmful gross chromosomal rearrangements
Homologous recombination is an essential process of DNA repair to maintain genomic integrity of the organism. Now, researchers from Japan have identified mechanisms that choose between alternate pathways of DNA repair to limit anomalous and deleterious chromosomal combinations that may be predisposed to cancer and genetic diseases.
In a recent study, researchers from Osaka University show that Rad52-dependent single-strand annealing (SSA) is the mechanism of homologous pairing that leads to gross chromosomal rearrangements (GCRs) at the centromere. They also identify mutations that allow this pathway to predominate in preference to the error-free Rad51 pathway.
Cells are under constant genotoxic pressure from exogenous and endogenous factors. Genome instability underlies several diseases including cancer. Consequently, DNA repair necessarily occurs thousands of times per day in each human cell to correct detrimental mutations, blockage of replication and transcription, and chromosomal breakage. Paradoxically, this recombination may occasionally cause dysfunctional GCRs.
The centromere of a chromosome is the specialized DNA sequence that links a pair of sister chromatids. Many organisms, including humans and fission yeast ( Schizosaccharomyces pombe ), have repetitive sequences at centromeres. This renders them vulnerable to isochromosome formation —dysfunctional mirror-images—due to a specific type of recombination between inverted repeats. In experiments conducted on fission yeast, the researchers demonstrated that DNA replication mechanisms reduce the occurrence of GCRs by inhibiting SSA activity at centromeres.
“At centromeres, Rad51-dependent recombination predominates and other recombination pathways appear to be inhibited,” explains Atsushi T. Onaka, lead author. “As Rad51 promotes conservative non-crossover recombination, the choice of recombination pathways is important for suppressing GCRs. This recombination predominantly occurs between inverted repeats, thereby suppressing formation of isochromosomes. However, how Rad51-dependent recombination predominates at centromeres is unknown.”
Jie Su, co-lead author, explains further. “We showed that Rad52-dependent SSA is the mechanism of homologous pairing that leads to centromeric GCRs. The rad52-R45K mutation impairs SSA activity of the Rad52 protein and reduces isochromosome formation in rad51 mutant cells. To better understand how Rad52-dependent SSA is suppressed at centromeres, we performed a genetic screen and found that specific mutations in replication fork proteins and a fork protection complex increase Rad52-dependent SSA at centromeres and isochromosome formation.”
“Our research implicates DNA replication machinery in the recombination pathway choice at centromeres, preventing Rad52-dependent SSA that results in GCRs,” explains senior author Takuro Nakagawa. “This knowledge identifies Rad52 as a promising target in treating cancer.”
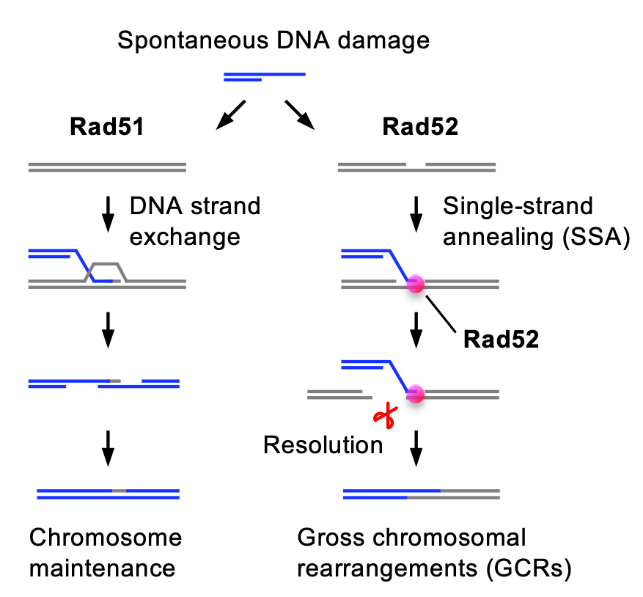
Fig. 1 Two pathways of DNA damage repair. Rad51 faithfully repairs DNA damage through DNA strand exchange with intact DNA (left). Rad52 causes gross chromosomal rearrangements (GCRs) through single-strand annealing (SSA) (right).
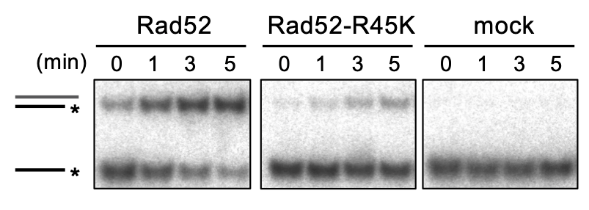
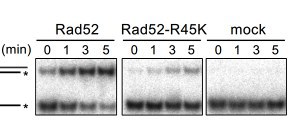
Fig. 2 An in vitro single-strand annealing (SSA) reaction. Compared to the wild-type Rad52, the mutant Rad52-R45K produced limited amounts of the SSA product (upper bands). *, 32 P-label.
The article, “DNA replication machinery prevents Rad52-dependent single-strand annealing that leads to gross chromosomal rearrangements at centromeres,” was published in Communications Biology at DOI: https://doi.org/10.1038/s42003-020-0934-0
Related Links
