
Patient registrations are now open at RUDY JAPAN website
RUDY JAPAN collects data directly from patients with rare diseases through self-completed questionnaires via web-based platform
A research team at Osaka University developed the Japanese version of the Rare and Undiagnosed Diseases Study (RUDY), RUDY JAPAN. Through collaboration with researchers at Hiroshima University, they began to accept the registration of hereditary angioedema (HAE) patients from October 30, 2018. Registered patients can fill out questionnaires on the RUDY JAPAN website [ https://rudy.hosp.med.osaka-u.ac.jp ] about their history of HAE attacks, which were newly prepared through cooperation between patients and researchers.
RUDY, a patient-driven medical research project using information and communication technology (ICT), was originally developed by the University of Oxford. The researchers at Osaka University introduced the RUDY platform in Japanese, called RUDY JAPAN. They began registration of patients with skeletal muscle channelopathies for the RUDY Japan Health Study in December 2017.
The use of the Internet and digital devices, such as personal computers and smartphones, has become increasingly more common. It facilitates the patients’ voluntary participation in medical studies using ICT, mainly in the U.K. and the U.S. However, this type of research in which not only researchers, but also patients, participate has rarely been conducted in Japan. Moreover, there were very few patients with rare/intractable diseases, so it was difficult to pursue studies at one medical or research institute.
RUDY JAPAN is a research project for collecting information of patients with rare/intractable diseases and advancing their research. The project offers an online research platform for gathering information about conditions and daily lives of patients. The RUDY JAPAN system includes three sections: (1) Profile, (2) To do, and (3) Timeline.
(1) On the “Profile” menu, registered patients can update their information, alter their consent in terms of use, and access their data and participation in secondary studies.
(2) On the “To do” menu, patients can answer questionnaires about drowsiness, activities of daily life, disease-specific quality of life, and degree of severity.
(3) On the “Timeline” menu, patients can refer to their completed questionnaires.
RUDY JAPAN aims for a new type of medical research to build research partnerships with patients/citizens. Thus, they hold regular online meetings, exchanging opinions with patients regarding the improvement of their medical research system and how to proceed with their research in the future.
Research team leader Prof. Kazuto Kato says, “I think we will be able to obtain detailed information about patients’ conditions and how these conditions affect their daily lives by connecting with patients across the nation through ICT. The use of RUDY JAPAN will allow long-term research on rare/intractable diseases. In addition, patients’ voluntary participation in research projects will reflect their voices on medical studies, sharing the objectives and benefitting studies.”

Fig.1: RUDY JAPAN web site [ https://rudy.hosp.med.osaka-u.ac.jp ].
(Osaka University)
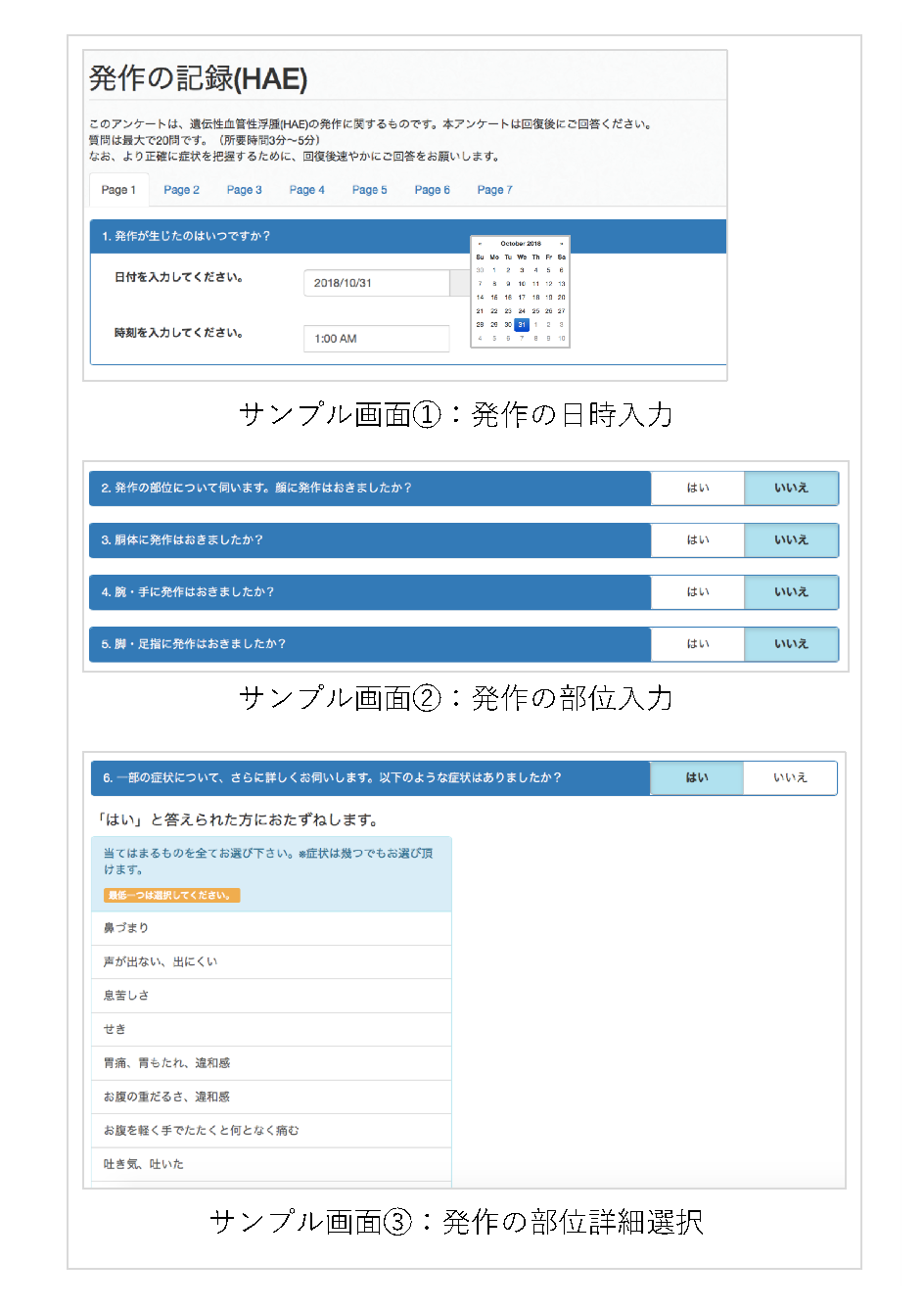
Fig. 2: Sample images of new questionnaires for hereditary angioedema (HAE) attacks
(credit: Osaka University)
Related links

