2018 MEXT Minister Prize for Science and Technology awarded for the development of asymmetric catalysts with unique activation mechanism
A group of researchers led by Professor Hiroaki SASAI at the Institute of Scientific and Industrial Research, Osaka University previously developed multi-point control type (dual activation type) asymmetric catalysts, in which multiple functional groups comprising the catalyst work together. This activation mechanism, which is similar to that of enzymes as biological catalysts, has enabled reactions that are not available through other catalysts.
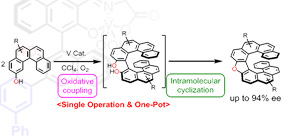
In this study, employing the concept of this activation mechanism, the researchers created dinuclear vanadium catalysts and acid-base organic catalysts without metals, applying them to cascade reactions in which sequential reactions take place. This has allowed them to synthesize oxahelicenes, which are not available through existing reactions, and other compounds that constitute basic skeletons of medicines.
The development of catalytic asymmetric synthesis is crucial for realizing a sustainable society. However, there are still many challenges in developing a catalytic asymmetric carbon-carbon bond-forming reaction, which is effective for making molecular skeletons of desired chemical compounds.
Ordinary catalysts selectively activate only one reactive partner in intramolecular reactions; however, the researchers developed catalysts that activate both reaction partners. They introduced this concept to metal-free catalysts, succeeding in cascade reactions in which multiple reactions take place in sequence.
This group’s achievements will lead to the development of new medical resources and organic electronics materials.
Figure 1
Related link
