Structural basis for RIFIN-mediated activation of LILRB1 in malaria
The Plasmodium species that cause malaria are obligate intracellular parasites, and disease symptoms occur as they replicate within human blood. Despite risking immune detection, the parasite delivers proteins that bind host receptors to infected erythrocyte surfaces. In the causative agent of the most deadly human malaria, Plasmodium falciparum, RIFINs form the largest erythrocyte surface protein family (Fig. 1).
Some RIFINs can bind inhibitory immune receptors, acting as targets for unusual antibodies containing a LAIR1 ectodomain, or as ligands for LILRB1. RIFINs potentially dampen human immune responses against malaria.
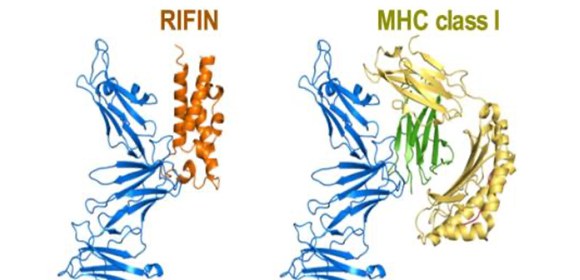
The research group of University of Oxford and Osaka University (led by Hisahi Arase, Immunochemistry, IFReC/RIMD) determined a structure of a RIFIN bound to LILRB1 (Fig. 2). They showed that the RIFIN mimics the natural activating ligand of LILRB1, MHC class I, in its LILRB1-binding mode. Therefore, LILRB1-binding RIFINs suppress NK cell function.

Fig.1 Mechanism of immune escape by P. falciparum (Saito et al. Nature 2017).

Fig. 2 Binding structure of LILRB1-binding RIFIN (L) and LILRB1-binding MHC I (R).
The article, "Structural basis for RIFIN-mediated activation of LILRB1 in malaria," is published in Nature at: https://www.nature.com/articles/s41586-020-2530-3 .
Related Links
