Small Metabolites Have Big Effects on the Intestinal Immune Response
Researchers at Osaka University find that common bacterial metabolites pyruvate and lactate enhance the intestinal immune response and guard against infection by important gut pathogens
For at least a decade, researchers have known that normal bacteria in the gut can induce intestinal immune cells to extend tentacle-like structures, known as dendrites, to “capture” antigens, triggering both immediate and long-term immune responses. What was less clear was how the bacteria activate this process. Now, a research team led by Osaka University has found that the molecules responsible have been hiding in plain sight.
Metabolites are small molecules produced during metabolism, the chemical processes that occur inside all living cells to keep them ticking over. Metabolic pathways have been intensively studied in many organisms, with most common metabolites having very few secrets. Yet, in a recent paper published in the journal Nature , the team describes how they made an important discovery—two well-known metabolites, pyruvate and lactate, are in fact the instigators of dendrite protrusion by CX3CR1+ macrophages in the small intestine.
“After studying the available research, we hypothesized that bacterial metabolites present in the small intestine could possibly mediate dendrite protrusion,” explains lead author Naoki Morita. “After purifying different fractions from the contents of the small intestines of mice, we discovered that lactic acid and pyruvic acid, produced by lactic acid bacteria in the normal gut flora, act directly on intestinal macrophages.”
Next, the researchers identified GPR31, a protein residing on the surface of small intestinal macrophages, as the specific receptor for the two metabolites. Mice lacking GPR31 showed reduced dendrite protrusion by CX3CR1+ cells after being administered pyruvate or lactate and, as a result, decreased antibody production following infection with a non-pathogenic strain of Salmonella . However, the most significant revelation was yet to come.
“We then examined whether pre-treatment of normal mice with pyruvate or lactate as well as non-pathogenic Salmonella could protect against infection with a virulent strain of the bacterium,” says co-lead author Eiji Umemoto. “As we predicted, normal pre-treated mice, but not pre-treated GPR31-defective mice, showed increased survival and an enhanced immune response following infection with the virulent Salmonella strain.”
Corresponding author Kiyoshi Takeda explains that the research has multiple clinical applications. “Because these metabolites enhance the immune response, they could be used to improve the effectiveness of oral vaccines, while GPR31 is a promising target for therapies aimed at eliminating intestinal pathogens. Because of this, we expect that lactic acid, pyruvic acid, and GPR31 will all be explored in the near future as new targets for activating immunity.”
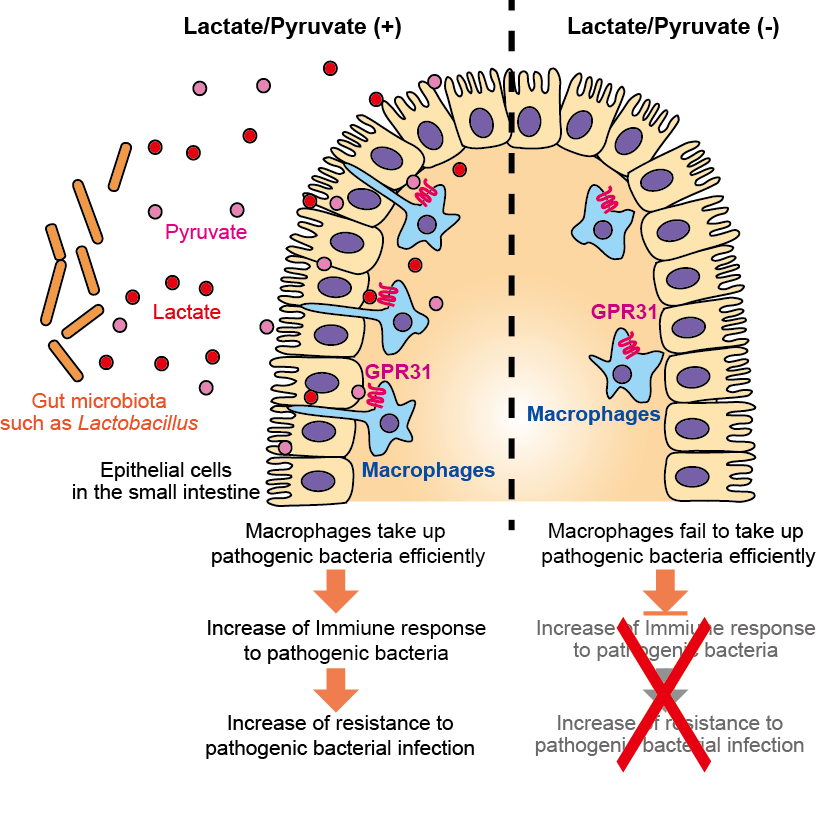
Fig. 1: Role of bacterial metabolites lactate and pyruvate in intestinal immune response.
Gut microbiota such as lactobacillus produce lactate and pyruvate. These metabolites stimulate intestinal macrophages through the receptor GPR31, allowing macrophages to protrude trans-epithelial dendrites and take up pathogenic bacteria efficiently in the intestine. Accordingly, lactate and pyruvate cause enhanced immune responses to pathogenic bacteria and increased resistance to the infection.
(Credit: Osaka University)
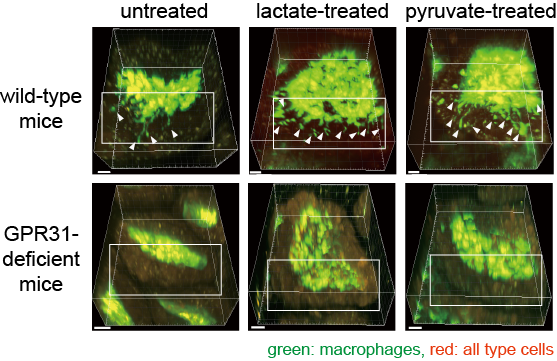
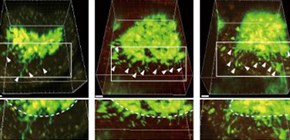
Fig. 2: Dendrite protrusion of intestinal macrophages by lactate and pyruvate.
Mice were orally administered with lactate or pyruvate for three weeks. 3D images of small intestinal villi were shown. Macrophages were marked in green, and their trans-epithelial dendrites were indicated by arrows. Administration of lactate or pyruvate dramatically increased the number of dendrites of macrophages in wild-type mice, but not in GPR31-deficient mice.
(Credit: Osaka University)
The article, “GPR31-dependent dendrite protrusion of intestinal CX3CR1+ cells by bacterial metabolites,” was published in Nature at DOI: https://doi.org/10.1038/s41586-019-0884-1 .
Related links
