Resist! TAK1 enables endothelial cells to avoid apoptosis
Osaka University-led study shows that TAK1 preserves endothelial cell survival in an inflammatory environment, and can be targeted for tumor suppression
Cell death is an important aspect of tissue homeostasis, as well as inflammation and disease pathogenesis related to infection, injury, and tumor growth. Tumor necrosis factor-α (TNFα) coordinates cell death in a variety of tissues; however, endothelial cells within blood vessels show a unique resistance to the action of TNFα. This resistance has been poorly understood, until now.
In a new study published in Developmental Cell , a research team led by experts from Osaka University investigated the mechanism underlying endothelial cell resistance to TNFα-induced apoptosis, an important mechanism of cell death in inflammatory conditions. They found that TAK1, a known signaling messenger involved in tissue homeostasis, was essential for survival of endothelial cells upon exposure to TNFα.
TNFα-induced cell death is tightly regulated and resisted in endothelial cells, which use TNFα exposure as a signal to exit the stationary “resting state” and become active and start inflammation. Despite a variety of research efforts, this resistance mechanism has remained ambiguous. Here, the researchers used a unique type of mouse, in which TAK1 could be deleted in specific tissues, to show that TAK1 is an essential component in ensuring survival of endothelial cells during inflammation and injury.
“We found that deletion of TAK1 blocked the ability of intestinal endothelial cells to disregard monocyte and macrophage inflammatory TNFα signaling induced by intestinal microbiota, resulting in hemorrhage within the intestine and liver,” says corresponding author Nobuyuki Takakura. “Thus, the presence of TAK1 safeguarded the survival of endothelial cells in these tissues.”
In the study, in vivo experiments with inflammation model mouse revealed that TAK1 was essential for survival of endothelial cells in lung tissue and hind limb muscle, under conditions similar to pneumonia and myositis.
“We suspected that inflammation in tumor tissues may be a powerful weapon to destroy tumor vasculature,” says lead author Hisamichi Naito. “We found that, in tumor in which TNFα is expressed abundantly, localized deletion of TAK1 resulted endothelial cell death and thereby tumor regression, suggesting that TAK1 may be a useful target in anti-angiogenic therapy.”
In addition to providing a clear understanding of endothelial cell survival during inflammation, this study has revealed a potential pathway for other cell types to avoid the onset of apoptosis, and may be important in future regenerative medicine therapies.
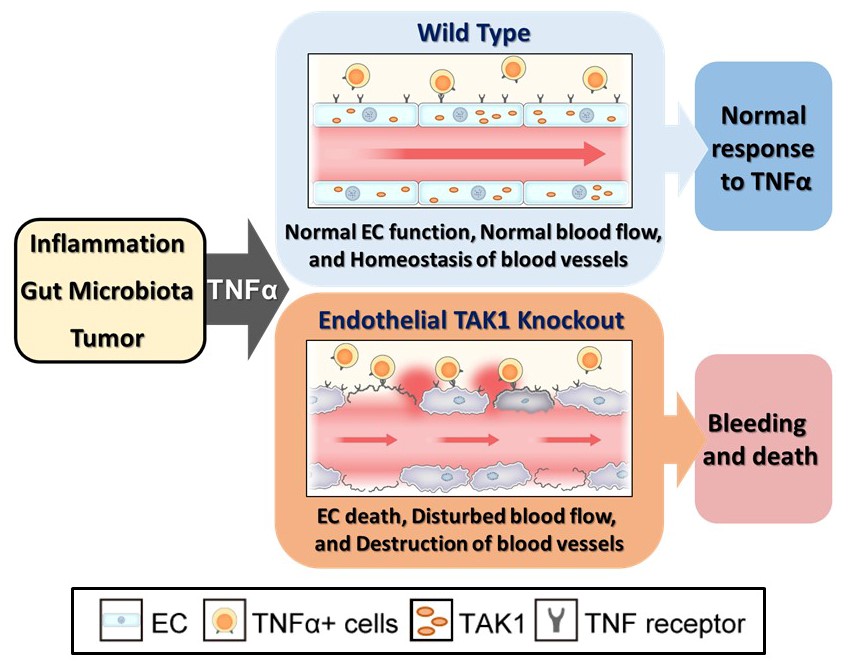
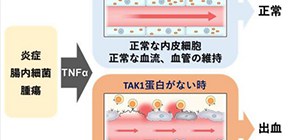
Fig. 1. TAK1 mechanism
The mechanism of vascular maintenance by endothelial TAK1. TAK1 maintains vascular integrity of the blood vessels by ensuring endothelial cell (EC) survival against TNFα induction by inflammation, gut microbe activity, or tumor presence. TAK1 deficiency results in EC death, leading to vascular dysfunction, hemorrhage, and rapid death of the animal.
(Credit: Osaka University)
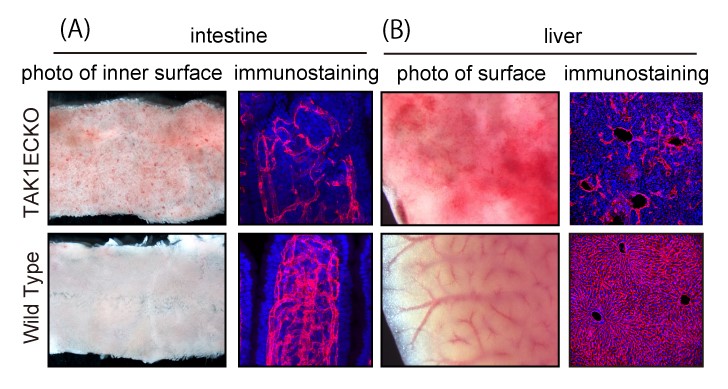
Fig. 2. TAK1ECKO results
Endothelial TAK1 deficiency results in hemorrhage and endothelial cell (EC) loss in the intestine and liver of adult mice.
(A) Brightfield and immunostaining images of small intestine. Intramucosal bleeding is observed in endothelial TAK1-deficient (TAK1ECKO) mice.
(B) Brightfield and immunostaining images of liver. Hepatic bleeding is observed in TAK1ECKO mice.
(A and B) Immunostaining images show that endothelial cells (red) of intestine and liver are destroyed in TAK1ECKO mice.
(©️ Naito et al., 2019, Developmental Cell 48, 1–16. DOI: 10.1016/j.devcel.2018.12.002)
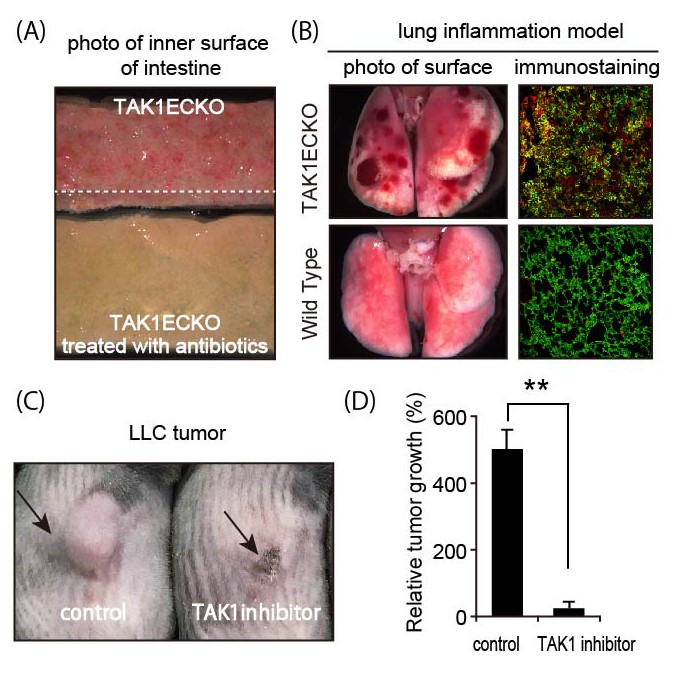
Fig.3
Gut microbes and inflammation induce vascular destruction and hemorrhage in endothelial TAK1-deficient (TAK1ECKO) mice. TAK1 inhibition causes tumor regression.
(A) Intestine of TAK1ECKO mouse with or without antibiotics. Intestinal bleeding was attenuated with antibiotics.
(B) Lungs of TAK1ECKO and wild-type mice showing acute inflammation. Destruction of blood vessels and hemorrhage occurred in TAK1ECKO mice. Immunostaining shows induction of TNFα (red) by acute inflammation and destruction of endothelial cells (green) in TAK1ECKO mice.
(C) Lewis lung carcinoma (LLC) tumor in a wild-type mouse, treated with TAK1 inhibitor.
(D) Tumor volume in (C) before and after treatment with TAK1 inhibitor.
(©️ Naito et al., 2019, Developmental Cell 48, 1–16. DOI: 10.1016/j.devcel.2018.12.002)
The article, "TAK1 Prevents Endothelial Apoptosis and Maintains Vascular Integrity," was published in Developmental Cell , at DOI: https://doi.org/10.1016/j.devcel.2018.12.002 .
Related links
