
Ultrathin black phosphorus for solar-driven hydrogen economy
Osaka University researchers use sunlight to make hydrogen with a new nanostructured catalyst based on nanosheets of black phosphorus and bismuth vanadate
Hydrogen as a fuel source, rather than hydrocarbons like oil and coal, offers many benefits. Burning hydrogen produces harmless water with the potential to eliminate carbon dioxide emissions and their environmental burden. In pursuit of technologies that could lead to a breakthrough in achieving a hydrogen economy, a key issue is making hydrogen cheaply. Using catalysts to split water is the ideal way to generate hydrogen, but doing so usually requires an energy input from other chemicals, electricity, or a portion of sunlight which has high enough energy.
Now researchers at Osaka University have developed a new catalytic system for efficiently splitting water and making hydrogen with energy from normal sunlight. Their study was recently reported in Angewandte Chemie International Edition .
“It has not been possible to use visible light for photocatalysis, but our approach of combining nanostructured black phosphorus for water reduction to hydrogen and bismuth vanadate for water oxidation to oxygen lets us make use of a wide range of the solar spectrum to make hydrogen and oxygen with unprecedented efficiency,” lead author Mingshan Zhu says.
Black phosphorus has a flat, two-dimensional structure similar to that of graphene and strongly absorbs light across the whole of the visible spectrum. The researchers combined the black phosphorus with bismuth vanadate, which is a well-known water oxidation catalyst.
In the same way that plants shuttle electrons between different structures in natural photosynthesis to split water and make oxygen, the two components of this new catalyst could rapidly transfer electrons excited by sunlight. The amounts of the two components was also optimized in the catalyst, leading to production of hydrogen and oxygen gases in an ideal 2:1 ratio.
Coauthor Tetsuro Majima says, “The realization of hydrogen production powered by sunlight is the foundation of a hydrogen-oriented society. Our contribution overcomes a significant hurdle, but much work remains to be done to make hydrogen a practical fuel source in the future.”
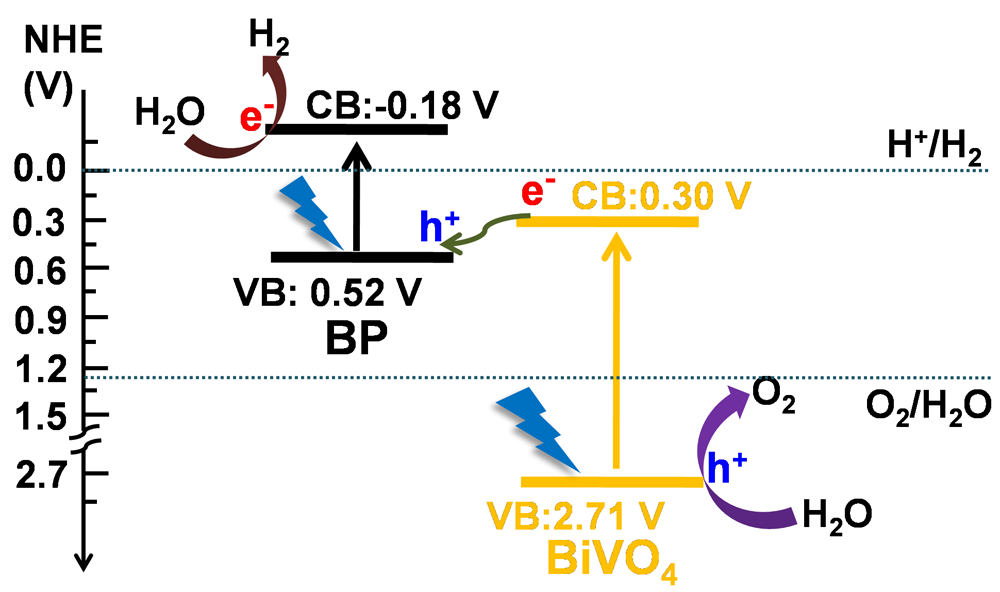
Fig.1. Schematic diagram of Z-scheme photocatalytic overall water splitting using BP/BiVO 4 under visible light irradiation.
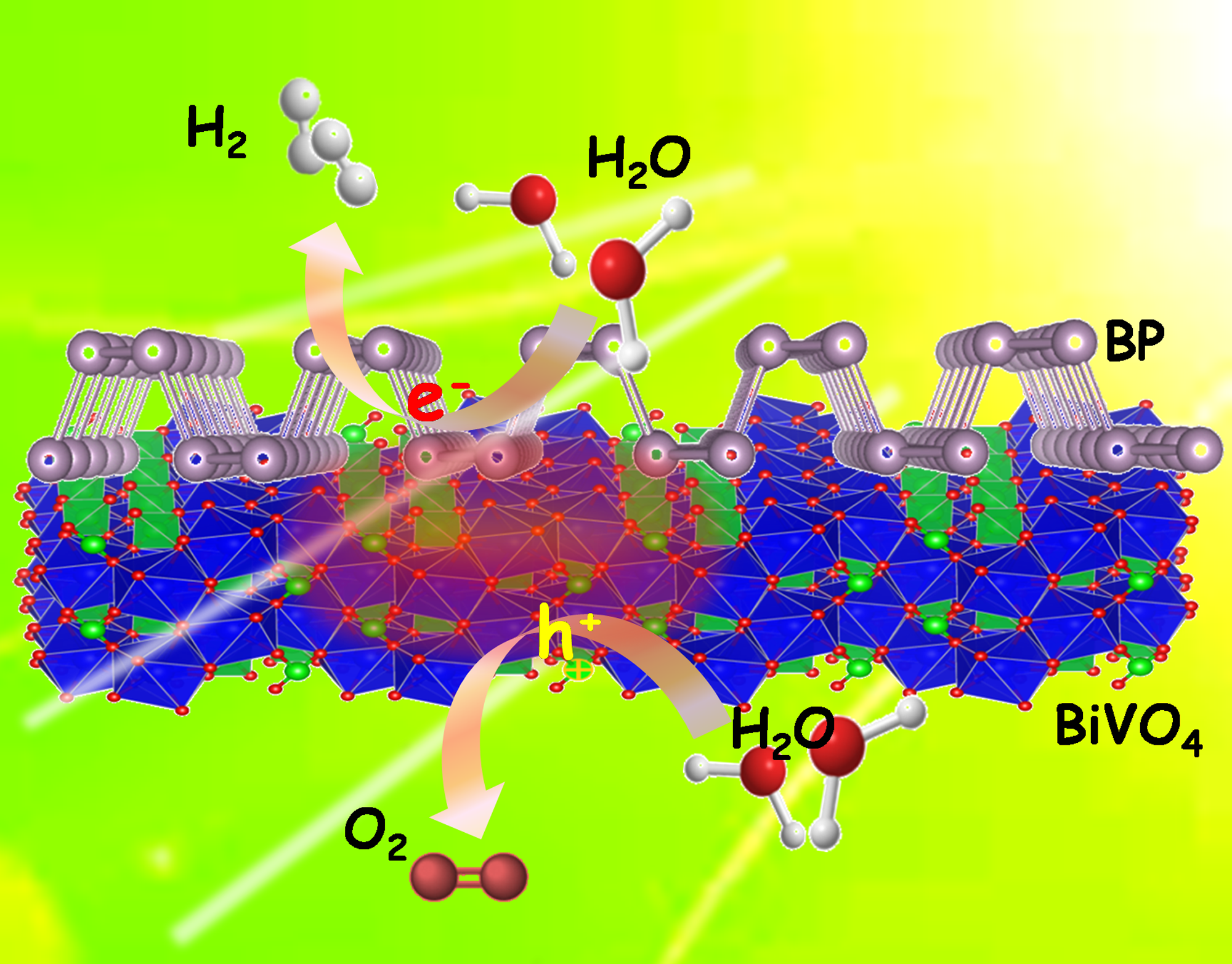
Fig.2. Photocatalytic overall pure-water splitting using the 2D heterostructures of BP/BiVO 4 without any sacrificial agents under visible light irradiation.
To learn more about this research, please view the full research report entitled " Z-Scheme Photocatalytic Overall Pure-Water Splitting on 2D Heterostructure of Black Phosphorus/BiVO₄ under Visible Light " at this page of Angewandte Chemie International Edition .
Related links
