Success in maintaining low blood pressure in hypertensive mice for 6 months
One step closer to a therapeutic vaccine for high blood pressure
Research Overview
A research team led by Assistant Professor KORIYAMA Hiroshi and Professor NAKAGAMI Hironori of the Department of Health Development and Medicine, Osaka University Graduate School of Medicine, and Professor MORISHITA Ryuichi of the Department of Clinical Gene Therapy, Graduate School of Medicine, Osaka University, with the aim of developing a therapeutic vaccine for hypertension, have successfully developed a DNA vaccine which targets Angiotensin II, a molecule which has been shown to increase blood pressure. Recently, there has been clinical testing overseas to treat hypertension through the use of vaccines targeting Angiotensin II, but the effects only lasted for a few weeks. However, the vaccine developed from this research, when given to mice with high blood pressure, maintained a lowered blood pressure for at least half of a year. As a result, the life-span of the rats in the administered group was also significantly lengthened. The creation of a long-lasting vaccine like the one found in these medical results may bring us closer to realizing a permanent cure for hypertension.
With the success of this research, it’s assumed that the dosing interval will become much longer than already existing antihypertensive medication, which could contribute to a significant decrease in necessary antihypertensive medication, an increase in adherence, a decrease in cardiovascular illness, and a reduction in medical costs. Looking forward, development will continue on the formulation of medication that will more efficiently exhibit these effects, with clinical trials already being planned.
Abstract
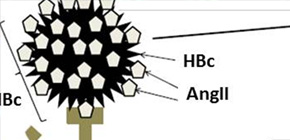
Recent research on vaccination has extended its scope from infectious diseases to chronic diseases, including Alzheimer disease, dyslipidemia, and hypertension. The aim of this study was to design DNA vaccines for high blood pressure and eventually develop human vaccine therapy to treat hypertension. Plasmid vector encoding hepatitis B core-angiotensin II (Ang II) fusion protein was injected into spontaneously hypertensive rats using needleless injection system.
Anti-Ang II antibody was successfully produced in hepatitis B core-Ang II group, and antibody response against Ang II was sustained for at least 6 months. Systolic blood pressure was consistently lower in hepatitis B core-Ang II group after immunization, whereas blood pressure reduction was continued for at least 6 months. Perivascular fibrosis in heart tissue was also significantly decreased in hepatitis B core-Ang II group. Survival rate was significantly improved in hepatitis B core-Ang II group. This study demonstrated that Ang II DNA vaccine to spontaneously hypertensive rats significantly lowered high blood pressure for at least 6 months. In addition, Ang II DNA vaccines induced an adequate humoral immune response while avoiding the activation of self-reactive T cells, assessed by ELISPOT assay. Future development of DNA vaccine to treat hypertension may provide a new therapeutic option to treat hypertension.
AngII : Angiotensin II, HBc : Hepatitis B Core, APC : Antigen Presenting Cell, CTL : Cytotoxic T Cell, Ang-N : Angiotensinogen
Plasmid vector encoding hepatitis B core -angiotensin II fusion protein was injected into spontaneously hypertensive rats. Then B cells produced anti-Angiotensin II antibody. As a result, serum angiotensin II concentration was decreased. Consequently, blood pressure reduction was continued for at least 6 months. On the other hand, AngII is very small and did not act as a T cell epitope. Therefore angiotensin II DNA vaccines did not induce the activation of self-reactive cytotoxic T cells (cytotoxic autoimmune reaction).
To learn more about this research, please view the full research report entitled " Long-Term Reduction of High Blood Pressure by Angiotensin II DNA Vaccine in Spontaneously Hypertensive Rats " at this page of the Hypertention website.
Related link

