Important role played by the "useless" appendix clarified
The appendix generates IgA and plays role in regulation of intestinal bacterial flora
A group of researchers from Osaka University, The University of Tokyo, Kinki University, and Riken have found that lymph tissue in the appendix, commonly misconceived as an unnecessary organ, was an important location for generating IgA that plays a crucial role in mucosal immunity and that it was involved in the regulation of intestinal bacterial flora.
Osaka University -- TAKEDA Kiyoshi , Professor, Laboratory of Immune Regulation, Office of Combined Program on Microbiology and Immunology, Graduate School of Medicine; Immunology Frontier Research Center
This group used germ-free mice, some of which were appendectomized. They found that in the appendectomized mice the number of IgA-producing cells in the large intestine was reduced and balance of intestinal bacterial flora was changed. IgA is an important antibody for maintaining a balance in intestinal bacterial flora. Thus, they demonstrated that the appendix is an important organ in the suppression of inflammatory bowel disease caused by a disturbed balance of intestinal bacterial flora.
This group also examined the structure of lymph tissue in the appendix by comparing it with Peyer's patch cells in the intestinal tract-related lymph tissue. In order to examine the function of lymph tissue in the appendix, this group removed the appendix from the germ-free mice with non-fully developed immunity and colonized intestinal bacterium. It was found that in the appendectomized mice, the increase in IgA-producing cells in the large intestine was significantly delayed.
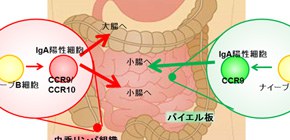
IgA is crucial in maintaining the balance in intestinal bacterial flora. This group examined the intestinal bacterial flora in both mice with an appendix and in appendectomized mice. Appendectomized mice did not show the large-intestine type pattern of intestinal bacterial flora. This group also found that while IgA-positive cells in lymph tissue in the appendix migrated to both the small intestine and large intestine, most IgA-positive cells in Peyer's patch cells migrated to small intestine.
Thus, they were able to show that removal of the appendix caused an imbalance in intestinal bacterial flora in the large intestine. Recently it has also been reported that changes in intestinal bacterial flora increase sensitivity to enteric infection and development of inflammatory bowel disease. The development of methods for regulating gut immunity flora that take into account the importance of the lymph tissue in the appendix may lead to the possible treatment of inflammatory bowel disease and other enteric infections.
Abstract
Gut-associated lymphoid tissues are responsible for the generation of IgA-secreting cells. However, the function of the caecal patch, a lymphoid tissue in the appendix, remains unknown. Here we analyse the role of the caecal patch using germ-free mice colonized with intestinal bacteria after appendectomy. Appendectomized mice show delayed accumulation of IgA + cells in the large intestine, but not the small intestine, after colonization. Decreased colonic IgA + cells correlate with altered faecal microbiota composition. Experiments using photoconvertible Kaede-expressing mice or adoptive transfer show that the caecal patch IgA + cells migrate to the large and small intestines, whereas Peyer’s patch cells are preferentially recruited to the small intestine. IgA + cells in the caecal patch express higher levels of CCR10 . Dendritic cells in the caecal patch, but not Peyer’s patches, induce CCR10 on cocultured B cells. Thus, the caecal patch is a major site for generation of IgA-secreting cells that migrate to the large intestine.


Figure 1
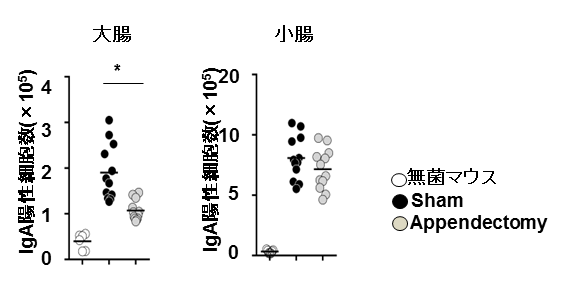
Figure 2
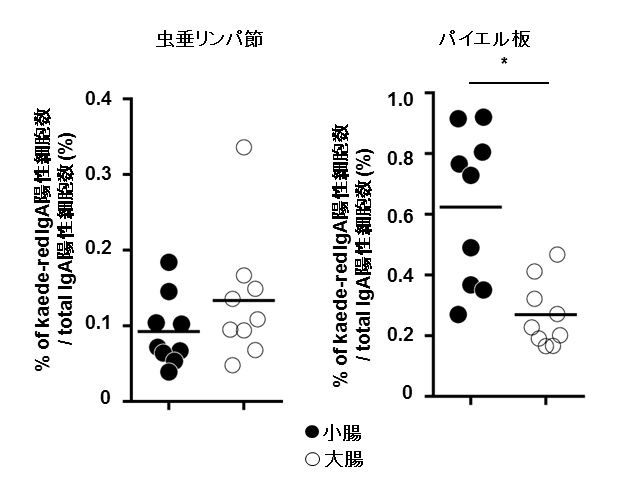
Figure 3
To learn more about this research, please read the full research report entitled "Generation of colonic IgA-secreting cells in the cecal patch" at this page of the Nature Communications website.
Related link :
